Entrez 2492 | Ensembl ENSG00000170820 | |
 | ||
Aliases FSHR, FSHR1, FSHRO, LGR1, ODG1, follicle stimulating hormone receptor External IDs OMIM: 136435 MGI: 95583 HomoloGene: 117 GeneCards: FSHR | ||
The follicle-stimulating hormone receptor or FSH receptor (FSHR) is a transmembrane receptor that interacts with the follicle-stimulating hormone (FSH) and represents a G protein-coupled receptor (GPCR). Its activation is necessary for the hormonal functioning of FSH. FSHRs are found in the ovary, testis, and uterus.
Contents
FSHR gene
The gene for the FSHR is found on chromosome 2 p21 in humans. The gene sequence of the FSHR consists of about 2,080 nucleotides.
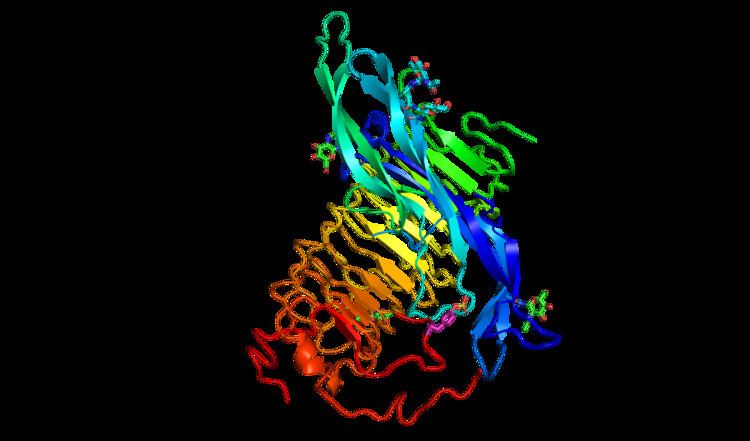
Receptor structure
The FSHR consists of 695 amino acids and has a molecular mass of about 76 kDa. Like other GPCRs, the FSH-receptor possesses seven membrane-spanning domains or transmembrane helices.
Ligand binding and signal transduction
Upon initial binding to the LRR region of FSHR, FSH reshapes its conformation to form a new pocket. FSHR then inserts its sulfotyrosine from the hinge loop into the pockets and activates the 7-helical transmembrane domain. This event leads to a transduction of the signal that activates the G protein that is bound to the receptor internally. With FSH attached, the receptor shifts conformation and, thus, mechanically activates the G protein, which detaches from the receptor and activates the cAMP system.
It is believed that a receptor molecule exists in a conformational equilibrium between active and inactive states. The binding of FSH to the receptor shifts the equilibrium between active and inactive receptors. FSH and FSH-agonists shift the equilibrium in favor of active states; FSH antagonists shift the equilibrium in favor of inactive states. For a cell to respond to FSH, only a small percentage (~1%) of receptor sites need to be activated.
Phosphorylation by cAMP-dependent protein kinases
Cyclic AMP-dependent protein kinases (protein kinase A) are activated by the signal chain coming from the G protein (that was activated by the FSH-receptor) via adenylate cyclase and cyclic AMP (cAMP).
These protein kinases are present as tetramers with two regulatory units and two catalytic units. Upon binding of cAMP to the regulatory units, the catalytic units are released and initiate the phosphorylation of proteins, leading to the physiologic action. The cyclic AMP-regulatory dimers are degraded by phosphodiesterase and release 5’AMP. DNA in the cell nucleus binds to phosphorylated proteins through the cyclic AMP response element (CRE), which results in the activation of genes.
The signal is amplified by the involvement of cAMP and the resulting phosphorylation. The process is modified by prostaglandins. Other cellular regulators are participate are the intracellular calcium concentration modified by phospholipase, nitric acid, and other growth factors.
The FSH receptor can also activate the extracellular signal-regulated kinases (ERK). In a feedback mechanism, these activated kinases phosphorylate the receptor. The longer the receptor remains active, the more kinases are activated, the more receptors are phosphorylated.
Action
In the ovary, the FSH receptor is necessary for follicular development and expressed on the granulosa cells.
In the male, the FSH receptor has been identified on the Sertoli cells that are critical for spermatogenesis.
The FSHR is expressed during the luteal phase in the secretory endometrium of the uterus.
FSH receptor is selectively expressed on the surface of the blood vessels of a wide range of carcinogenic tumors.
Upregulation
Upregulation refers to the increase in the number of receptor site on the membrane. Estrogen upregulates FSH receptor sites. In turn, FSH stimulates granulosa cells to produce estrogens. This synergistic activity of estrogen and FSH allows for follicle growth and development in the ovary.
Desensitization
The FSHR become desensitized when exposed to FSH for some time. A key reaction of this downregulation is the phosphorylation of the intracellular (or cytoplasmic) receptor domain by protein kinases. This process uncouples Gs protein from the FSHR. Another way to desensitize is to uncouple the regulatory and catalytic units of the cAMP system.
Downregulation
Downregulation refers to the decrease in the number of receptor sites. This can be accomplished by metabolizing bound FSHR sites. The bound FSH-receptor complex is brought by lateral migration to a "coated pit," where such units are concentrated and then stabilized by a framework of clathrins. A pinched-off coated pit is internalized and degraded by lysosomes. Proteins may be metabolized or the receptor can be recycled. Use of long-acting agonists will downregulate the receptor population.
Modulators
Antibodies to FSHR can interfere with FSHR activity.
FSH abnormalities
Some patients with ovarian hyperstimulation syndrome may have mutations in the gene for FSHR, making them more sensitive to gonadotropin stimulation.
Women with 46 XX gonadal dysgenesis experience primary amenorrhea with hypergonadotropic hypogonadism. There are forms of 46 xx gonadal dysgenesis wherein abnormalities in the FSH-receptor have been reported and are thought to be the cause of the hypogonadism.
Polymorphism may affect FSH receptor populations and lead to poorer responses in infertile women receiving FSH medication for IVF.
History
Alfred G. Gilman and Martin Rodbell received the 1994 Nobel Prize in Medicine and Physiology for the discovery of the G Protein System.
