Group Group V ((-)ssRNA) Rank Genus | Scientific name Henipavirus | |
 | ||
Similar Flying foxes, Megabat, Morbillivirus, Lyssavirus, Australian bat lyssavirus | ||
Henipavirus is a genus of RNA viruses in the family Paramyxoviridae, order Mononegavirales containing five established species. Henipaviruses are naturally harboured by pteropid fruit bats (flying foxes) and microbats of several species. Henipaviruses are characterised by long genomes and a wide host range. Their recent emergence as zoonotic pathogens capable of causing illness and death in domestic animals and humans is a cause of concern.
Contents
- Taxonomy
- Virus structure
- Genome structure
- Emergence
- Transmission
- Australian outbreaks
- Events of JuneAugust 2011
- Prevention detection and treatment
- Pathology
- 2016 Brisbane Research Conference
- Evolution
- Outbreaks
- Causes of emergence
- References
In 2009, RNA sequences of three novel viruses in phylogenetic relationship to known henipaviruses were detected in African straw-colored fruit bats (Eidolon helvum) in Ghana. The finding of these novel henipaviruses outside Australia and Asia indicates that the region of potential endemicity of henipaviruses may be worldwide. These African henipaviruses are slowly being characterised.
Taxonomy
Table legend: "*" denotes type species.
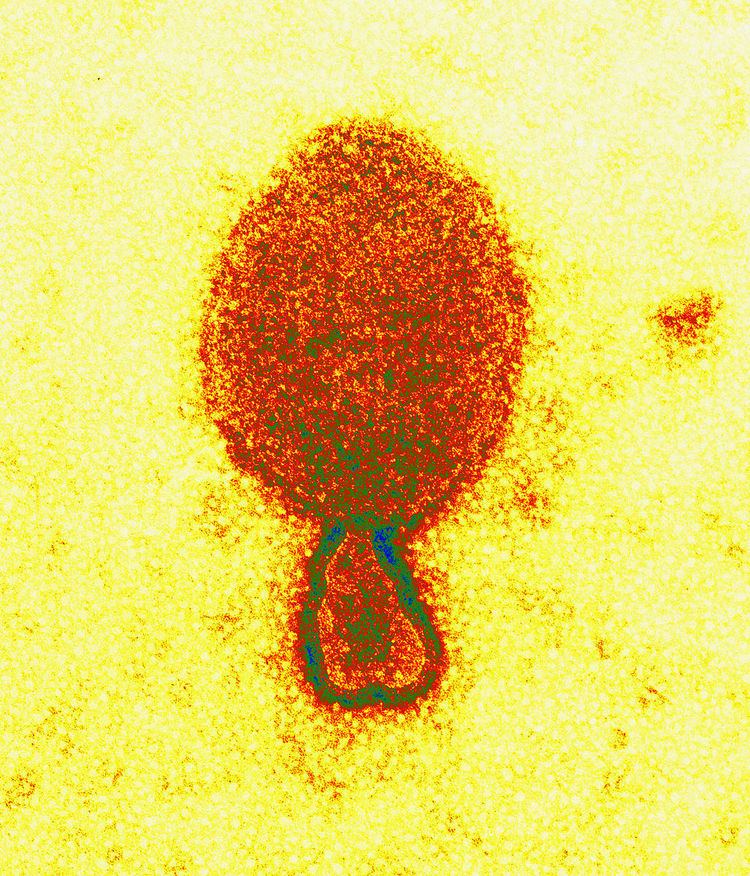
Virus structure
Henipaviriona are pleomorphic (variably shaped), ranging in size from 40 to 600 nm in diameter. They possess a lipid membrane overlying a shell of viral matrix protein. At the core is a single helical strand of genomic RNA tightly bound to N (nucleocapsid) protein and associated with the L (large) and P (phosphoprotein) proteins, which provide RNA polymerase activity during replication.
Embedded within the lipid membrane are spikes of F (fusion) protein trimers and G (attachment) protein tetramers. The function of the G protein is to attach the virus to the surface of a host cell via EFNB2, a highly conserved protein present in many mammals. The structure of the attachment glycoprotein has been determined by X-ray crystallography. The F protein fuses the viral membrane with the host cell membrane, releasing the virion contents into the cell. It also causes infected cells to fuse with neighbouring cells to form large, multinucleated syncytia.
Genome structure
As all mononegaviral genomes, Hendra virus and Nipah virus genomes are non-segmented, single-stranded negative-sense RNA. Both genomes are 18.2 kb in length and contain six genes corresponding to six structural proteins.
In common with other members of the Paramyxoviridae family, the number of nucleotides in the henipavirus genome is a multiple of six, consistent with what is known as the 'rule of six'. Deviation from the rule of six, through mutation or incomplete genome synthesis, leads to inefficient viral replication, probably due to structural constraints imposed by the binding between the RNA and the N protein.
Henipaviruses employ an unusual process called RNA editing to generate multiple proteins from a single gene. The specific process in henipaviruses involves the insertion of extra guanosine residues into the P gene mRNA prior to translation. The number of residues added determines whether the P, V or W proteins are synthesised. The functions of the V and W proteins are unknown, but they may be involved in disrupting host antiviral mechanisms.
Emergence
Hendra virus (originally called "Equine morbillivirus" was discovered in September 1994 when it caused the deaths of thirteen horses, and a trainer at a training complex in Hendra, a suburb of Brisbane in Queensland, Australia.
The index case, a mare called Drama Series, was housed with 19 other horses after falling ill, and died two days later. Subsequently, all of the horses became ill, with 13 dying. The remaining six animals were subsequently euthanised as a way of preventing relapsing infection and possible further transmission. The trainer, Victory ('Vic') Rail, and the stable foreman, Ray Unwin, were involved in nursing the index case, and both fell ill with an influenza-like illness within one week of the first horse’s death. The stable hand recovered while Rail died of respiratory and renal failure. The source of the virus was most likely frothy nasal discharge from the index case.
A second outbreak occurred in August 1994 (chronologically preceding the first outbreak) in Mackay 1,000 km north of Brisbane resulting in the deaths of two horses and their owner. The owner, Mark Preston, assisted in necropsies of the horses and within three weeks was admitted to hospital suffering from meningitis. Mr Preston recovered, but 14 months later developed neurologic signs and died. This outbreak was diagnosed retrospectively by the presence of Hendra virus in the brain of the patient.
Transmission
Flying foxes have been identified as the reservoir host of Hendra Virus. A seroprevalence of 47% is found in the flying foxes, suggesting an endemic infection of the bat population throughout Australia. Horses become infected with Hendra after exposure to bodily fluid from an infected flying fox. This often happens in the form of urine, feces, or masticated fruit covered in the flying fox's saliva when horses are allowed to graze below roosting sites. The seven human cases have all been infected only after contact with sick horses. As a result, veterinarians are particularly at risk for contracting the disease.
There is no evidence of bat to human, human to human, or human to horse transmission.
Australian outbreaks
As of June 2014, a total of fifty outbreaks of Hendra virus have occurred in Australia, all involving infection of horses. As a result of these events, eighty-three horses have died or been euthanised. A further four died or were euthanised as a result of possible hendra infection.
Case fatality rate in humans is 60% and in horses 75%.
Four of these outbreaks have spread to humans as a result of direct contact with infected horses. On 26 July 2011 a dog living on the Mt Alford property was reported to have HeV antibodies, the first time an animal other than a flying fox, horse, or human has tested positive outside an experimental situation.
These events have all been on the east coast of Australia, with the most northern event at Cairns, Queensland and the event furthest south at Kempsey, NSW. Until the event at Chinchilla, Queensland in July 2011, all outbreak sites had been within the distribution of at least two of the four mainland flying foxes (fruit bats); Little red flying fox, (Pteropus scapulatus), black flying fox, (Pteropus alecto), grey-headed flying fox, (Pteropus poliocephalus) and spectacled flying fox, (Pteropus conspicillatus). Chinchilla is considered to be only within the range of little red flying fox and is west of the Great Dividing Range. This is the furthest west the infection has ever been identified in horses.
The timing of incidents indicates a seasonal pattern of outbreaks. Initially this was thought to possibly related to the breeding cycle of the little red flying foxes. These species typically give birth between April and May. Subsequently however, the Spectacled flying fox and the Black flying fox have been identified as the species more likely to be involved in infection spillovers.
Timing of outbreaks also appears more likely during the cooler months when it is possible the temperature and humidity are more favourable to the longer term survival of the virus in the environment.
There is no evidence of transmission to humans directly from bats, and, as such it appears that human infection only occurs via an intermediate host, a horse. Despite this in 2014 the NSW Government sanctioned the destruction of flying fox colonies.
Events of June–August 2011
In the years 1994–2010, fourteen events were recorded. Between 20 June 2011 and 28 August 2011, a further seventeen events were identified, during which twenty-one horses died.
It's not clear why there has been a sudden increase in the number of spillover events between June and August 2011. Typically HeV spillover events are more common between May and October. This time is sometimes called "Hendra Season", which is a time when there are large numbers of fruit bats of all species congregated in SE Queensland's valuable winter foraging habitat. The weather (warm and humid) is favourable to the survival of henipavirus in the environment.
It is possible flooding in SE Queensland and Northern NSW in December 2010 and January 2011 may have affected the health of the fruit bats. Urine sampling in flying fox camps indicate that a larger proportion of flying foxes than usual are shedding live virus. Biosecurity Queensland's ongoing surveillance usually shows 7% of the animals are shedding live virus. In June and July nearly 30% animals have been reported to be shedding live virus. Present advice is that these events are not being driven by any mutation in HeV itself.
Other suggestions include that an increase in testing has led to an increase in detection. As the actual mode of transmission between bats and horses has not been determined, it is not clear what, if any, factors can increase the chance of infection in horses.
Following the confirmation of a dog with HeV antibodies, on 27 July 2011, the Queensland and NSW governments will boost research funding into the Hendra virus by $6 million to be spent by 2014–2015. This money will be used for research into ecological drivers of infection in the bats and the mechanism of virus transmission between bats and other species. A further 6 million dollars was allocated by the federal government with the funds being split, half for human health investigations and half for animal health and biodiversity research.
Prevention, detection and treatment
Three main approaches are currently followed to reduce the risk to humans.
Pathology
Flying foxes experimentally infected with the Hendra virus develop a viraemia then excrete the virus in their urine, faeces and saliva for approximately one week. Although they excrete active virus during this time there is no other indication of an illness. Symptoms of Hendra virus infection of humans may be respiratory, including hemorrhage and edema of the lungs, or encephalitic, resulting in meningitis. In horses, infection usually causes pulmonary oedema, congestion and / or neurological signs.
Ephrin B2 has been identified as the main receptor for the henipaviruses.
Hendra virus has been classified as a Bio-safety Level 4 Hot Agent.
2016 Brisbane Research Conference
In late May, early June 2016, "Hendra scientists, vets and health officers met in Brisbane to bring together all the research into the bat-borne disease."
Emergence
Nipah virus was identified in April 1999, when it caused an outbreak of neurological and respiratory disease on pig farms in peninsular Malaysia, resulting in 257 human cases, including 105 human deaths and the culling of one million pigs. In Singapore, 11 cases, including one death, occurred in abattoir workers exposed to pigs imported from the affected Malaysian farms. The Nipah virus has been classified by the Centers for Disease Control and Prevention as a Category C agent. The name "Nipah" refers to the place, Kampung Baru Sungai Nipah in Port Dickson, Negeri Sembilan, the source of the human case from which Nipah virus was first isolated. Nipah virus is one of several viruses identified by WHO as a likely cause of a future epidemic in a new plan developed after the Ebola epidemic for urgent research and development before and during an epidemic toward new diagnostic tests, vaccines and medicines.
The outbreak was originally mistaken for Japanese encephalitis (JE), however, physicians in the area noted that persons who had been vaccinated against JE were not protected, and the number of cases among adults was unusual Despite the fact that these observations were recorded in the first month of the outbreak, the Ministry of Health failed to react accordingly, and instead launched a nationwide campaign to educate people on the dangers of JE and its vector, Culex mosquitoes.
Symptoms of infection from the Malaysian outbreak were primarily encephalitic in humans and respiratory in pigs. Later outbreaks have caused respiratory illness in humans, increasing the likelihood of human-to-human transmission and indicating the existence of more dangerous strains of the virus.
Based on seroprevalence data and virus isolations, the primary reservoir for Nipah virus was identified as Pteropid fruit bats, including Pteropus vampyrus (Large Flying Fox), and Pteropus hypomelanus (Small flying fox), both of which occur in Malaysia.
The transmission of Nipah virus from flying foxes to pigs is thought to be due to an increasing overlap between bat habitats and piggeries in peninsular Malaysia. At the index farm, fruit orchards were in close proximity to the piggery, allowing the spillage of urine, faeces and partially eaten fruit onto the pigs. Retrospective studies demonstrate that viral spillover into pigs may have been occurring in Malaysia since 1996 without detection. During 1998, viral spread was aided by the transfer of infected pigs to other farms, where new outbreaks occurred.
Evolution
The most likely origin of this virus was in 1947 (95% credible interval: 1888-1988). There are two clades of this virus - one with its origin in 1995 (95% credible interval: 1985-2002) and a second with its origin in 1985 (95% credible interval: 1971-1996). The mutation rate was estimated to be 6.5 × 10−4 substitution/site/year (95% credible interval: 2.3 × 10−4 -1.18 × 10−3) similar to other RNA viruses.
Outbreaks
Eight more outbreaks of Nipah virus have occurred since 1998, all within Bangladesh and neighbouring parts of India. The outbreak sites lie within the range of Pteropus species (Pteropus giganteus). As with Hendra virus, the timing of the outbreaks indicates a seasonal effect. Cases occurring in Bangladesh during the winters of 2001, 2003, and 2004, were determined to have been caused by the Nipah virus. In February 2011, a Nipah outbreak began at Hatibandha Upazila in the Lalmonirhat District of northern Bangladesh. To date (7 February 2011), there have been 24 cases and 17 deaths in this outbreak.
Nipah virus has been isolated from Lyle's flying fox (Pteropus lylei) in Cambodia and viral RNA found in urine and saliva from P. lylei and Horsfield's roundleaf bat (Hipposideros larvatus) in Thailand. Infective virus has also been isolated from environmental samples of bat urine and partially eaten fruit in Malaysia. Antibodies to henipaviruses have also been found in fruit bats in Madagascar (Pteropus rufus, Eidolon dupreanum) and Ghana (Eidolon helvum) indicating a wide geographic distribution of the viruses. No infection of humans or other species have been observed in Cambodia, Thailand or Africa thus far.
Pathology
In humans, the infection presents as fever, headache and drowsiness. Cough, abdominal pain, nausea, vomiting, weakness, problems with swallowing and blurred vision are relatively common. About a quarter of the patients have seizures and about 60% become comatose and might need mechanical ventilation. In patients with severe disease, their conscious state may deteriorate and they may develop severe hypertension, fast heart rate, and very high temperature.
Nipah virus is also known to cause relapse encephalitis. In the initial Malaysian outbreak, a patient presented with relapse encephalitis some 53 months after his initial infection. There is no definitive treatment for Nipah encephalitis, apart from supportive measures, such as mechanical ventilation and prevention of secondary infection. Ribavirin, an antiviral drug, was tested in the Malaysian outbreak, and the results were encouraging, though further studies are still needed.
While no vaccine currently exists, a recent (2012) study of a trial vaccine developed using the outer proteins of Hendra virus was shown to induce protection against Nipah in African Green Monkeys.
In animals, especially in pigs, the virus causes a porcine respiratory and neurologic syndrome, locally known as "barking pig syndrome" or "one mile cough."
Ephrin B2 has been identified as the main receptor for the henipaviruses.
Emergence
Cedar Virus (CedV) was first identified in pteropid urine during work on Hendra virus undertaken in Queensland in 2009.
Although the virus is reported to be very similar to both Hendra and Nipah viruses, it does not cause illness in laboratory animals usually susceptible to paramyxoviruses. Animals were able to mount an effective response and create effective antibodies.
The scientists who identified the virus report:
Hendra and Nipah viruses are 2 highly pathogenic paramyxoviruses that have emerged from bats within the last two decades. Both are capable of causing fatal disease in both humans and many mammal species. Serological and molecular evidence for henipa-like viruses have been reported from numerous locations including Asia and Africa, however, until now no successful isolation of these viruses have been reported. This paper reports the isolation of a novel paramyxovirus, named Cedar virus, from fruit bats in Australia. Full genome sequencing of this virus suggests a close relationship with the henipaviruses. Antibodies to Cedar virus were shown to cross react with, but not cross neutralize Hendra or Nipah virus. Despite this close relationship, when Cedar virus was tested in experimental challenge models in ferrets and guinea pigs, we identified virus replication and generation of neutralizing antibodies, but no clinical disease was observed. As such, this virus provides a useful reference for future reverse genetics experiments to determine the molecular basis of the pathogenicity of the henipaviruses.Causes of emergence
The emergence of henipaviruses parallels the emergence of other zoonotic viruses in recent decades. SARS coronavirus, Australian bat lyssavirus, Menangle virus and probably Ebola virus and Marburg virus are also harbored by bats and are capable of infecting a variety of other species. The emergence of each of these viruses has been linked to an increase in contact between bats and humans, sometimes involving an intermediate domestic animal host. The increased contact is driven both by human encroachment into the bats’ territory (in the case of Nipah, specifically pigpens in said territory) and by movement of bats towards human populations due to changes in food distribution and loss of habitat.
There is evidence that habitat loss for flying foxes, both in South Asia and Australia (particularly along the east coast) as well as encroachment of human dwellings and agriculture into the remaining habitats, is creating greater overlap of human and flying fox distributions.
