Formula C7H6O Molar mass 106.121 g/mol Melting point -26 °C | Density 1.04 g/cm³ Boiling point 178.1 °C Soluble in Water | |
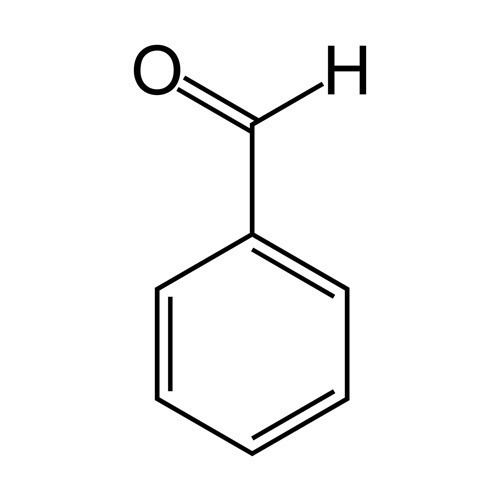 | ||
Related compounds Appearance colorless or yellowish liquid; strongly refractive | ||
Benzaldehyde from bitter almond oil
Benzaldehyde (C6H5CHO) is an organic compound consisting of a benzene ring with a formyl substituent. It is the simplest aromatic aldehyde and one of the most industrially useful.
Contents
- Benzaldehyde from bitter almond oil
- History
- Production
- Occurrence
- Reactions
- Uses
- Niche uses
- Safety
- References
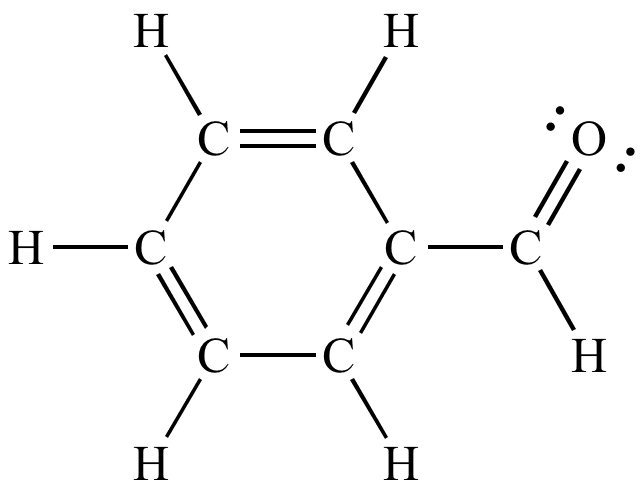
This colorless liquid has a characteristic almond-like odor. Benzaldehyde is the primary component of bitter almond oil and can be extracted from a number of other natural sources. Synthetic benzaldehyde is the flavoring agent in imitation almond extract, which is used to flavor cakes and other baked goods.
History
Benzaldehyde was first extracted from bitter almonds in 1803 by the French pharmacist Martrès. In 1832 German chemists Friedrich Wöhler and Justus von Liebig first synthesized benzaldehyde.
Production
Benzaldehyde can be obtained by many processes. In the 1980s, an estimated 18 million kilograms were produced annually in Japan, Europe, and North America, a level that can be assumed to continue. Currently liquid phase chlorination and oxidation of toluene are the main routes. Numerous other methods have been developed, such as the partial oxidation of benzyl alcohol, alkali hydrolysis of benzal chloride, and the carbonylation of benzene.
Benzaldehyde can be synthesized from cinnamaldehyde obtained from the oil of cinnamon by refluxing in aqueous/alcoholic solution between 90 °C and 150 °C with a base (most commonly sodium carbonate or bicarbonate) for 5 to 80 hours, followed by distillation of the formed benzaldehyde. This reaction also yields acetaldehyde.
"Site-specific nuclear magnetic resonance spectroscopy", which evaluates 1H/2H isotope ratios, has been used to differentiate between naturally occurring and synthetic benzaldehyde.
Occurrence
Benzaldehyde and similar chemicals occur naturally in many foods. Most of the benzaldehyde that people eat is from natural, traditional foods, such as almonds.
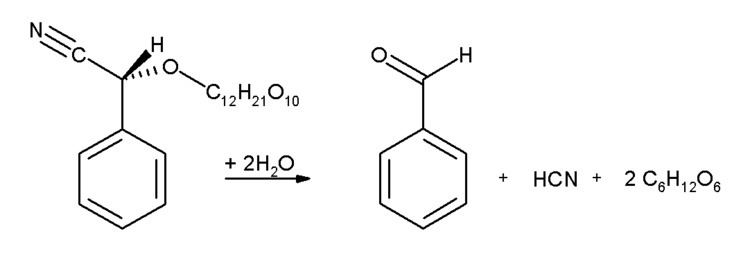
Almonds, apricots, apples and cherry kernels, contain significant amounts of amygdalin. This glycoside breaks up under enzyme catalysis into benzaldehyde, hydrogen cyanide and two molecules of glucose.
Benzaldehyde contributes to the scent of oyster mushrooms (Pleurotus ostreatus).
Reactions
On oxidation, benzaldehyde is converted into the odorless benzoic acid, which is a common impurity in laboratory samples. Benzyl alcohol can be formed from benzaldehyde by means of hydrogenation. Reaction of benzaldehyde with anhydrous sodium acetate and acetic anhydride yields cinnamic acid, while alcoholic potassium cyanide can be used to catalyze the condensation of benzaldehyde to benzoin. Benzaldehyde undergoes disproportionation upon treatment with concentrated alkali (Cannizzaro reaction): one molecule of the aldehyde is reduced to the corresponding alcohol and another molecule is simultaneously oxidized to benzoate.
Uses
Benzaldehyde is commonly employed to confer almond flavor to foods and scented products. It is sometimes used in cosmetics products.
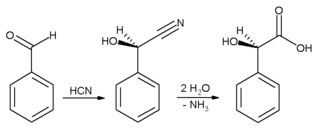
In industrial settings, benzaldehyde is used chiefly as a precursor to other organic compounds, ranging from pharmaceuticals to plastic additives. The aniline dye malachite green is prepared from benzaldehyde and dimethylaniline. It is a precursor to certain acridine dyes as well. Via aldol condensations, benzaldehyde is converted into derivatives of cinnamaldehyde and styrene. The synthesis of mandelic acid starts from benzaldehyde:
First hydrocyanic acid is added to benzaldehyde, and the resulting nitrile is subsequently hydrolysed to mandelic acid. (The scheme above depicts only one of the two formed enantiomers).
Niche uses
It is used as a bee repellant. A small amount of benzaldehyde-containing solution is placed on a fume board near the honey combs. The bees promptly move away from the honey combs to get away from the fumes. Benzaldehyde allows the beekeeper to remove the honey frames from the bee hive with greater safety to both bees and the beekeeper.
Safety
For a 70-kg human, the lethal dose is estimated at 50 mL. Benzaldehyde has been classified as a hazardous substance by the United States Environmental Protection Agency.
Benzaldehyde is used as a flavoring and fragrance in food, cosmetics, pharmaceuticals, and soap and is "generally regarded as safe" (GRAS) by the US FDA. This status was reaffirmed after a review in 2005. It is accepted in the European Union as a flavoring agent. Toxicology studies indicate that it is safe and non-carcinogenic in the concentrations used for foods and cosmetics. It may have carcinostatic (anti-cancer) properties.
Benzaldehyde does not accumulate in any specific tissues. It is metabolized and then excreted in urine.
