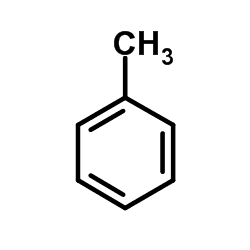
Abbreviations PhMeMePhBnH Related compounds Boiling point 110.6 °C Molar mass 92.14 g/mol ChEBI ID 17578 | Appearance Colorless liquid Formula C6H5-CH3 Density 867 kg/m³ Pubchem 1140 | |
 | ||
Thermodynamicdata Phase behavioursolid–liquid–gas | ||
Toluene exposure risks concerns
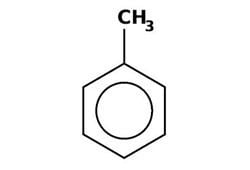
Toluene /ˈtɒljuːiːn/, also known as toluol /ˈtɒljuːɒl/, is a colorless, water-insoluble liquid with the smell associated with paint thinners. It is a mono-substituted benzene derivative, consisting of a CH3 group attached to a phenyl group. As such, its IUPAC systematic name is methylbenzene. It is an aromatic hydrocarbon.
Contents
- Toluene exposure risks concerns
- Fractional distillation of lacquer thinner to obtain toluene
- History
- Chemical properties
- Production
- Laboratory preparation
- Uses
- Precursor to other chemicals
- Solvent
- Fuel
- Niche applications
- Toxicology and metabolism
- Bioremediation
- Recreational drug use and legal status
- References
Toluene is widely used as an industrial feedstock and a solvent. In 2013, worldwide sales of toluene amounted to about 24.5 billion US-dollars.
As the solvent in some types of paint thinner, contact cement and model airplane glue, toluene is sometimes used as a recreational inhalant and has the potential of causing severe neurological harm.
Fractional distillation of lacquer thinner to obtain toluene
History
The compound was first isolated in 1837 through a distillation of pine oil by a Polish chemist named Filip Walter, who named it rétinnaphte. In 1841, French chemist Henri Étienne Sainte-Claire Deville isolated a hydrocarbon from balsam of Tolu (an aromatic extract from the tropical Colombian tree Myroxylon balsamum), which Deville recognized as similar to Walter's rétinnaphte and to benzene; hence he called the new hydrocarbon benzoène. In 1843, Jöns Jacob Berzelius recommended the name toluin. In 1850, French chemist Auguste Cahours isolated from a distillate of wood a hydrocarbon which he recognized as similar to Deville's benzoène and which Cahours named toluène.
Chemical properties
Toluene reacts as a normal aromatic hydrocarbon in electrophilic aromatic substitution. Because the methyl group has greater electron-releasing properties than a hydrogen atom in the same position, toluene is more reactive than benzene to electrophiles. It undergoes sulfonation to give p-toluenesulfonic acid, and chlorination by Cl2 in the presence of FeCl3 to give ortho and para isomers of chlorotoluene.
Importantly, the methyl side chain in toluene is susceptible to oxidation. Toluene reacts with Potassium permanganate to yield benzoic acid, and with chromyl chloride to yield benzaldehyde (Étard reaction).
The methyl group undergoes halogenation under free radical conditions. For example, N-bromosuccinimide (NBS) heated with toluene in the presence of AIBN leads to benzyl bromide. The same conversion can be effected with elemental bromine in the presence of UV light or even sunlight. Toluene may also be brominated by treating it with HBr and H2O2 in the presence of light.
C6H5CH3 + Br2 → C6H5CH2Br + HBrC6H5CH2Br + Br2 → C6H5CHBr2 + HBrThe methyl group in toluene undergoes deprotonation only with very strong bases, its pKa is estimated to be approximately 41. Catalytic hydrogenation of toluene gives methylcyclohexane. The reaction requires a high pressure of hydrogen and a catalyst.
Production
Toluene occurs naturally at low levels in crude oil and is a byproduct in the production of gasoline by a catalytic reformer or ethylene cracker; It is also a byproduct of the production of coke from coal. Final separation and purification is done by any of the distillation or solvent extraction processes used for BTX aromatics (benzene, toluene, and xylene isomers).
Laboratory preparation
Toluene is so inexpensively produced industrially that it is not prepared in the laboratory. In principle it could be prepared by a variety of methods. For example, although only of didactical interest, benzene reacts with methyl chloride in presence of a Lewis acid such as aluminium chloride to give toluene:
C6H5H + CH3Cl → C6H5CH3 + HClSuch reactions are complicated by polymethylation because toluene is more susceptible to alkylation than is benzene.
Uses
Toluene is mainly used as a precursor to benzene via hydrodealkylation:
C6H5CH3 + H2 → C6H6 + CH4The second ranked application involves its disproportionation to a mixture of benzene and xylene. When oxidized it yields benzaldehyde and benzoic acid, two important intermediates in chemistry.
Precursor to other chemicals
In addition to the synthesis of benzene and xylene, toluene is a feedstock for toluene diisocyanate (used in the manufacture of polyurethane foam), trinitrotoluene (the explosive, TNT), and a number of synthetic drugs.
Solvent
Toluene is a common solvent, e.g. for paints, paint thinners, silicone sealants, many chemical reactants, rubber, printing ink, adhesives (glues), lacquers, leather tanners, and disinfectants.
Fuel
Toluene can be used as an octane booster in gasoline fuels for internal combustion engines. Toluene at 86% by volume fueled all the turbo Formula One teams in the 1980s, first pioneered by the Honda team. The remaining 14% was a "filler" of n-heptane, to reduce the octane to meet Formula One fuel restrictions. Toluene at 100% can be used as a fuel for both two-stroke and four-stroke engines; however, due to the density of the fuel and other factors, the fuel does not vaporize easily unless preheated to 70 °C (158 °F). Honda solved this problem in their Formula One cars by routing the fuel lines through the exhaust system to heat the fuel.
In Australia in 2003, toluene was found to have been illegally combined with petrol in fuel outlets for sale as standard vehicular fuel. Toluene incurs no fuel excise tax, while other fuels are taxed at more than 40%, providing a greater profit margin for fuel suppliers. The extent of toluene substitution has not been determined.
Toluene is one of a group of fuels that have recently included in jet fuel surrogate blends. Toluene is used as a jet fuel surrogate for its content of aromatic compounds.
Niche applications
In the laboratory, toluene is used as a solvent for carbon nanomaterials, including nanotubes and fullerenes, and it can also be used as a fullerene indicator. The color of the toluene solution of C60 is bright purple. Toluene is used as a cement for fine polystyrene kits (by dissolving and then fusing surfaces) as it can be applied very precisely by brush and contains none of the bulk of an adhesive. Toluene can be used to break open red blood cells in order to extract hemoglobin in biochemistry experiments. Toluene has also been used as a coolant for its good heat transfer capabilities in sodium cold traps used in nuclear reactor system loops. Toluene had also been used in the process of removing the cocaine from coca leaves in the production of Coca-Cola syrup.
Toxicology and metabolism
Inhalation of toluene in low to moderate levels can cause tiredness, confusion, weakness, drunken-type actions, memory loss, nausea, loss of appetite, hearing loss, and color vision loss. These symptoms usually disappear when exposure is stopped. Inhaling high levels of toluene in a short time may cause light-headedness, nausea, or sleepiness, unconsciousness, and even death. Toluene is, however, much less toxic than benzene, and has as a consequence, largely replaced it as an aromatic solvent in chemical preparation. The US Environmental Protection Agency (EPA) states that the carcinogenic potential of toluene cannot be evaluated due to insufficient information.
Similar to many other solvents such as 1,1,1-trichloroethane and some alkylbenzenes, toluene has been shown to act as a non-competitive NMDA receptor antagonist and GABAA receptor positive allosteric modulator. Additionally, toluene has been shown to display antidepressant-like effects in rodents in the forced swim test (FST) and the tail suspension test (TST).
Toluene is sometimes used as a recreational inhalant ("glue sniffing"), likely on account of its euphoric and dissociative effects.
Bioremediation
Several types of fungi including Cladophialophora, Exophiala, Leptodontium, Pseudeurotium zonatum, and Cladosporium sphaerospermum, and certain species of bacteria can degrade toluene using it as a source of carbon and energy.
Recreational drug use and legal status
Toluene is used as an intoxicative inhalant in a manner not intended by manufacturers. People inhale toluene-containing products (e.g., paint thinner, contact cement, model glue, etc.) for its intoxicating effect. The possession and use of toluene and products containing it are regulated in many jurisdictions, to prevent minors from obtaining these products for recreational drug purposes. As of 2007, 24 U.S. states had laws penalizing use, possession with intent to use, and/or distribution of such inhalants. Intentionally inhaling toluene vapours can be harmful or fatal.
