Appearance Colorless liquid Density 1.04 g/cm³ Molar mass 108.14 g/mol Melting point -15 °C | Formula C7H8O Boiling point 205 °C IUPAC ID Phenylmethanol Soluble in Water | |
 | ||
Water and benzyl alcohol
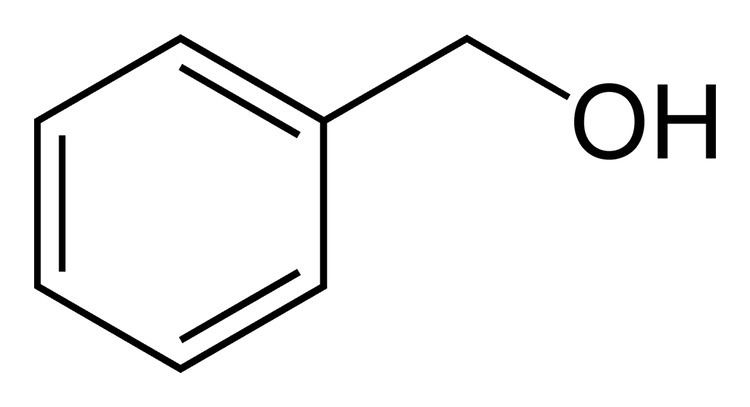
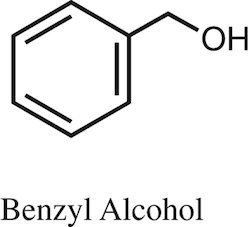
Benzyl alcohol is an aromatic alcohol with the formula C6H5CH2OH. The benzyl group is often abbreviated "Bn" (not to be confused with "Bz" which is used for benzoyl), thus benzyl alcohol is denoted as BnOH. Benzyl alcohol is a colorless liquid with a mild pleasant aromatic odor. It is a useful solvent due to its polarity, low toxicity, and low vapor pressure. Benzyl alcohol has moderate solubility in water (4 g/100 mL) and miscible in alcohols and diethyl ether. The anion produced by deprotonation of the alcohol group is known as benzylate or benzyloxide.
Contents
- Water and benzyl alcohol
- Natural occurrences
- Preparation
- Reactions
- Applications
- Nanotechnological uses
- Use in health care
- Other uses
- Safety
- References
Natural occurrences
Benzyl alcohol is produced naturally by many plants and is commonly found in fruits and teas. It is also found in a variety of essential oils including jasmine, hyacinth, and ylang-ylang.
It is also one of the chemical compounds found in castoreum from the castor sacs of beavers.
Preparation
Benzyl alcohol is prepared by the hydrolysis of benzyl chloride using sodium hydroxide:
C6H5CH2Cl + NaOH → C6H5CH2OH + NaCl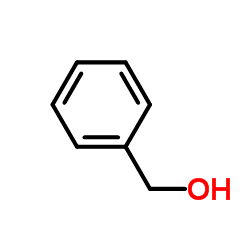
It can also be prepared via a Grignard reaction by reacting phenylmagnesium bromide (C6H5MgBr) with formaldehyde, followed by acidification.
Reactions
Like most alcohols, it reacts with carboxylic acids to form esters. In organic synthesis, benzyl esters are popular protecting groups because they can be removed by mild hydrogenolysis.
Benzyl alcohol reacts with acrylonitrile to give N-benzylacrylamide. This is an example of a Ritter reaction:
C6H5CH2OH + NCCHCH2 → C6H5CH2N(H)C(O)CHCH2Applications
Benzyl alcohol is used as a general solvent for inks, waxes, shellacs, paints, lacquers, and epoxy resin coatings. Thus it can be used in paint strippers, especially when combined with compatible viscosity enhancers to encourage the mixture to cling to painted surfaces. It is a precursor to a variety of esters, used in the soap, perfume, and flavor industries. It is also used in e-liquid for e-cigarettes to enhance the flavors used. When applied to damaged skin or mucous membranes at a 10% concentration it acts as a local anesthetic and antimicrobial agent, and is sometimes included as a degreaser in rug cleaning products. As a dye solvent, it enhances the process of dying wool, nylon, and leather. It also has use as a photographic developer, and as an insect repellent.
Nanotechnological uses
Benzyl alcohol has been used as a dielectric solvent for the dielectrophoretic reconfiguration of nanowires.
Use in health care
Benzyl alcohol is used as a bacteriostatic preservative at low concentration in intravenous medications, cosmetics and topical drugs.
The use of benzyl alcohol as a 5% solution has been approved by the U.S. FDA for the treatment of head lice in children older than 6 months and in adults. It affects the louse's spiracles, preventing them from closing. These then become clogged with water or mineral oil or other matter and cause the insect to die from asphyxiation.
Other uses
Benzyl alcohol has nearly the same refraction index as quartz and wool fiber. If a clear quartz object is immersed in benzyl alcohol, it becomes almost invisible. This test has been used to determine non-destructively whether an object is truly made of quartz (see crystal skull). Similarly, white wool immersed in benzyl alcohol also becomes almost invisible clearly revealing contaminants such as dark and medullated fibers and vegetable matter. Benzyl alcohol is used effectively for treating lice infestations as the active ingredient in lotion shampoo with 5% benzyl alcohol.
Safety
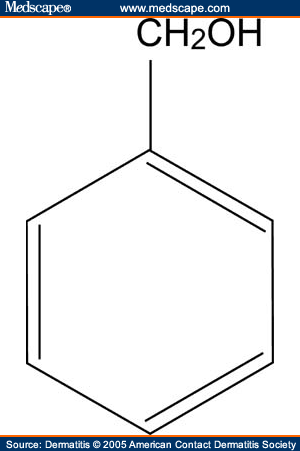
Benzyl alcohol has low acute toxicity with an LD50 of 1.2 g/kg in rats. It oxidizes rapidly in healthy individuals to benzoic acid, conjugated with glycine in the liver, and excreted as hippuric acid. Very high concentrations can result in toxic effects including respiratory failure, vasodilation, hypotension, convulsions, and paralysis.
Benzyl alcohol is toxic to neonates, it is associated with the gasping syndrome.
Benzyl alcohol has been reported to cause skin allergy.
Benzyl alcohol is severely toxic and highly irritating to the eye. Pure benzyl alcohol produces corneal necrosis.
Benzyl alcohol is not considered to be a carcinogen and no data are available regarding teratogenic or reproductive effects.
