Formula C3H4N2 Boiling point 256 °C | Molar mass 68.077 g/mol Density 1.23 g/cm³ | |
 | ||
Appearance white or pale yellow solid | ||
Imidazole
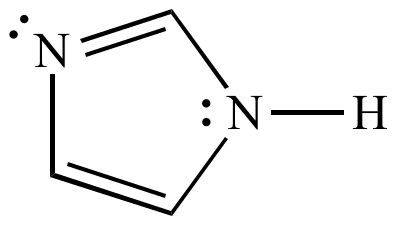
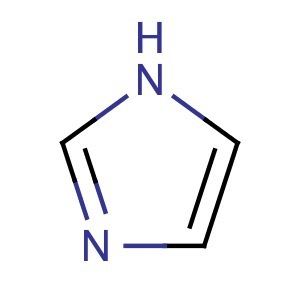
Imidazole is an organic compound with the formula C3N2H4. It is a white or colourless solid that is soluble in water, producing a mildly alkaline solution. In chemistry, it is an aromatic heterocycle, classified as a diazole, and having non-adjacent nitrogen atoms.
Contents
- Imidazole
- Structure and properties
- Amphoterism
- Preparation
- Biological significance and applications
- Pharmaceutical derivatives
- Industrial applications
- Salts of imidazole
- Related heterocycles
- References

Many natural products, especially alkaloids, contain the imidazole ring. These imidazoles share the 1,3-C3N2 ring but feature varied substituents. This ring system is present in important biological building blocks, such as histidine and the related hormone histamine. Many drugs contain an imidazole ring, such as certain antifungal drugs, the nitroimidazole series of antibiotics, and the sedative midazolam.
When fused to a pyrimidine ring, it forms purine, which is the most widely occurring nitrogen-containing heterocycle in nature.
The name "imidazole" was coined in 1887 by the German chemist Arthur Rudolf Hantzsch (1857–1935).
Structure and properties
Imidazole is a planar 5-membered ring. It exists in two equivalent tautomeric forms, because the positive charge can be located on either of the two nitrogen atoms. Imidazole is a highly polar compound, as evidenced by its electric dipole moment of 3.67 D. It is highly soluble in water. The compound is classified as aromatic due to the presence of a sextet of π-electrons, consisting of a pair of electrons from the protonated nitrogen atom and one from each of the remaining four atoms of the ring. Some resonance structures of imidazole are shown below:
Amphoterism
Imidazole is amphoteric. That is, it can function as both an acid and as a base. As an acid, the pKa of imidazole is 14.5, making it less acidic than carboxylic acids, phenols, and imides, but slightly more acidic than alcohols. The acidic proton is located on N-1. As a base, the pKa of the conjugate acid (cited as pKBH+ to avoid confusion between the two) is approximately 7, making imidazole approximately sixty times more basic than pyridine. The basic site is N3. Protonation gives the imidazolium cation, which is symmetrical.
Preparation
Imidazole was first reported in 1858 by the German-British chemist Heinrich Debus, although various imidazole derivatives had been discovered as early as the 1840s. Its synthesis, as shown below, used glyoxal and formaldehyde in ammonia to form imidazole (or glyoxaline, as it was originally named). This synthesis, while producing relatively low yields, is still used for creating C-substituted imidazoles.
In one microwave modification, the reactants are benzil, benzaldehyde and ammonia in glacial acetic acid, forming 2,4,5-triphenylimidazole (lophine).
Imidazole can be synthesized by numerous methods besides the Debus method. Many of these syntheses can also be applied to different substituted imidazoles and imidazole derivatives by varying the functional groups on the reactants. These methods are commonly categorized by which and how many bonds form to make the imidazole rings. For example, the Debus method forms the (1,2), (3,4), and (1,5) bonds in imidazole, using each reactant as a fragment of the ring, and thus this method would be a three-bond-forming synthesis. A small sampling of these methods is presented below.
Biological significance and applications
Imidazole is incorporated into many important biological molecules. The most pervasive is the amino acid histidine, which has an imidazole side-chain. Histidine is present in many proteins and enzymes and plays a vital part in the structure and binding functions of hemoglobin. Imidazole-based histidine compounds play a very important role in intracellular buffering. Histidine can be decarboxylated to histamine, which is also a common biological compound. It can cause urticaria (hives), when histamine is produced during allergic reaction. The relationship between histidine and histamine are shown below:
One of the applications of imidazole is in the purification of His-tagged proteins in immobilised metal affinity chromatography (IMAC). Imidazole is used to elute tagged proteins bound to nickel ions attached to the surface of beads in the chromatography column. An excess of imidazole is passed through the column, which displaces the His-tag from nickel coordination, freeing the His-tagged proteins.
Imidazole has become an important part of many pharmaceuticals. Synthetic imidazoles are present in many fungicides and antifungal, antiprotozoal, and antihypertensive medications. Imidazole is part of the theophylline molecule, found in tea leaves and coffee beans, that stimulates the central nervous system. It is present in the anticancer medication mercaptopurine, which combats leukemia by interfering with DNA activities.
A number of substituted imidazoles, including clotrimazole, are selective inhibitors of nitric oxide synthase, which makes them interesting drug targets in inflammation, neurodegenerative diseases and tumors of the nervous system. Other biological activities of the imidazole pharmacophore relate to the downregulation of intracellular Ca2+ and K+ fluxes, and interference with translation initiation.
Pharmaceutical derivatives
The substituted imidazole derivatives are valuable in treatment of many systemic fungal infections. Imidazoles belong to the class of azole antifungals, which includes ketoconazole, miconazole, and clotrimazole.
For comparison, another group of azoles is the triazoles, which includes fluconazole, itraconazole, and voriconazole. The difference between the imidazoles and the triazoles involves the mechanism of inhibition of the cytochrome P450 enzyme. The N3 of the imidazole compound binds to the heme iron atom of ferric cytochrome P450, whereas the N4 of the triazoles bind to the heme group. The triazoles have been shown to have a higher specificity for the cytochrome P450 than imidazoles, thereby making them more potent than the imidazoles.
Some imidazole derivatives show effects on insects, for example sulconazole nitrate exhibits a strong anti-feeding effect on the keratin-digesting Australian carpet beetle larvae Anthrenocerus australis, as does econazole nitrate with the common clothes moth Tineola bisselliella.
Industrial applications
Imidazole has been used extensively as a corrosion inhibitor on certain transition metals, such as copper. Preventing copper corrosion is important, especially in aqueous systems, where the conductivity of the copper decreases due to corrosion.
Many compounds of industrial and technological importance contain imidazole derivatives. The thermostable polybenzimidazole (PBI) contains imidazole fused to a benzene ring and linked to a benzene, and acts as a fire retardant. Imidazole can also be found in various compounds that are used for photography and electronics.
Salts of imidazole
Salts of imidazole where the imidazole ring is in the cation are known as imidazolium salts (for example, imidazolium chloride). These salts are formed from the protonation or substitution at nitrogen of imidazole. These salts have been used as ionic liquids and precursors to stable carbenes. Salts where a deprotonated imidazole is an anion are also well known; these salts are known as imidazolates (for example, sodium imidazolate, NaC3H3N2).
