Entrez 7273 | Ensembl ENSG00000155657 | |
 | ||
Aliases TTN, CMD1G, CMH9, CMPD4, EOMFC, HMERF, LGMD2J, MYLK5, TMD, titin External IDs MGI: 98864 HomoloGene: 130650 GeneCards: TTN | ||
Titin /ˈtaɪtɪn/, also known as connectin, is a protein that, in humans, is encoded by the TTN gene. Titin is a giant protein, greater than 1 µm in length, that functions as a molecular spring which is responsible for the passive elasticity of muscle. It is composed of 244 individually folded protein domains connected by unstructured peptide sequences. These domains unfold when the protein is stretched and refold when the tension is removed.
Contents
- Discovery
- Genomics
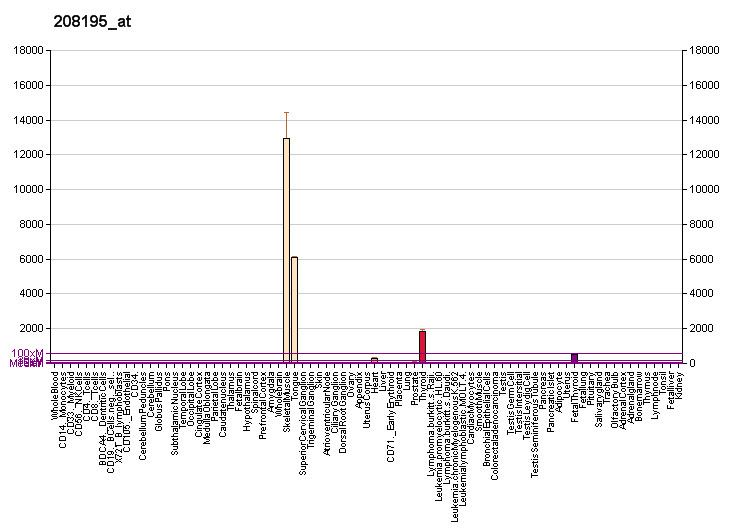
- Isoforms
- Structure
- Function
- Clinical relevance
- Interactions
- Linguistic significance
- References
Titin is important in the contraction of striated muscle tissues. It connects the Z line to the M line in the sarcomere. The protein contributes to force transmission at the Z line and resting tension in the I band region. It limits the range of motion of the sarcomere in tension, thus contributing to the passive stiffness of muscle. Variations in the sequence of titin between different types of muscle (e.g., cardiac or skeletal) have been correlated with differences in the mechanical properties of these muscles.
Titin is the third most abundant protein in muscle (after myosin and actin), and an adult human contains approximately 0.5 kg of titin. With its length of ~27,000 to ~33,000 amino acids (depending on the splice isoform), titin is the largest known protein. Furthermore, the gene for titin contains the largest number of exons (363) discovered in any single gene, as well as the longest single exon (17,106 bp).
Discovery
Reiji Natori in 1954 was the first to propose an elastic structure in muscle fiber to account for the return to the resting state when muscles are stretched and then released. In 1977, Koscak Maruyama and coworkers isolated an elastic protein from muscle fiber which they called connectin. Two years later, Kuan Wang and coworkers identified a doublet band on electrophoresis gel corresponding to a high molecular weight elastic protein which they named titin.
Labeit in 1990 isolated a partial cDNA clone of titin. In 1995, Labeit and Kolmerer determined the cDNA sequence of human cardiac titin. Bang and colleagues in 2001 determined the complete sequence of the human titin gene.
Genomics
The human gene encoding for titin is located on the long arm of chromosome 2 and contains 363 exons, which together code for 38,138 residues (4200 kDa). Within the gene are found a large number of PEVK exons 84 to 99 nucleotides in length which code for conserved 28- to 33-residue motifs which may represent structural units of the titin PEVK spring. The number of PEVK motifs in the titin gene appears to have increased during evolution, apparently modifying the genomic region responsible for titin’s spring properties.
Isoforms
A number of titin isoforms are produced in different striated muscle tissues as a result of alternative splicing. All but one of these isoforms are in the range of ~27,000 to ~36,000 amino acid residues in length. The exception is the small cardiac novex-3 isoform which is only 5,604 amino acid residues in length. The following table lists the known titin isoforms:
Structure
Titin is the largest known protein; its human variant consists of 34,350 amino acids, with the molecular weight of the mature "canonical" isoform of the protein being approximately 3,816,188.13 Da. Its mouse homologue is even larger, comprising 35,213 amino acids with a MW of 3,906,487.6 Da. It has a theoretical isoelectric point of 6.01. The protein's empirical chemical formula is C169,719H270,464N45,688O52,237S911. It has a theoretical instability index (II) of 42.41, classifying the protein as unstable. The protein's in vivo half-life, the time it takes for half of the amount of protein in a cell to break down after its synthesis in the cell, is predicted to be approximately 30 hours (in mammalian reticulocytes).
The titin protein is located between the myosin thick filament and the Z disk. Titin consists primarily of a linear array of two types of modules (also referred to as protein domains; 244 copies in total): type I (fibronectin type III domain; 132 copies) and type II (immunoglobulin domain; 112 copies). This linear array is further organized into two regions:
Function
Titin is a large abundant protein of striated muscle. Titin's primary functions are to stabilize the thick filament, center it between the thin filaments, prevent overstretching of the sarcomere, and to recoil the sarcomere like a spring after it is stretched. An N-terminal Z-disc region and a C-terminal M-line region bind to the Z-line and M-line of the sarcomere, respectively, so that a single titin molecule spans half the length of a sarcomere. Titin also contains binding sites for muscle-associated proteins so it serves as an adhesion template for the assembly of contractile machinery in muscle cells. It has also been identified as a structural protein for chromosomes. Considerable variability exists in the I-band, the M-line and the Z-disc regions of titin. Variability in the I-band region contributes to the differences in elasticity of different titin isoforms and, therefore, to the differences in elasticity of different muscle types. Of the many titin variants identified, five are described with complete transcript information available.
Titin interacts with many sarcomeric proteins including:
Clinical relevance
Mutations anywhere within the unusually long sequence of this gene can cause premature stop codons or other defects. Titin mutations are associated with hereditary myopathy with early respiratory failure, early-onset myopathy with fatal cardiomyopathy, core myopathy with heart disease, centronuclear myopathy, Limb-girdle muscular dystrophy type 2J, familial dilated cardiomyopathy 9, hypertrophic cardiomyopathy and tibial muscular dystrophy. Further research also suggests that no genetically linked form of any dystrophy or myopathy can be safely excluded from being caused by a mutation on the TTN gene. Truncating mutations in dilated cardiomyopathy patients are most commonly found in the A region; although truncations in the upstream I region might be expected to prevent translation of the A region entirely, alternative splicing creates some transcripts that do not encounter the premature stop codon, ameliorating its effect.
Autoantibodies to titin are produced in patients with the autoimmune disease scleroderma.
Interactions
Titin has been shown to interact with:
Linguistic significance
The name titin is derived from the Greek Titan (a giant deity, anything of great size).
As the largest known protein, titin also has the longest IUPAC name of a protein. The full chemical name of the human canonical form of titin, which starts methionyl... and ends ...isoleucine, contains 189,819 letters and is sometimes stated to be the longest word in the English language, or any language. However, lexicographers regard generic names of chemical compounds as verbal formulae rather than English words.
