 | ||
Polyketide synthases (PKSs) are a family of multi-domain enzymes or enzyme complexes that produce polyketides, a large class of secondary metabolites, in bacteria, fungi, plants, and a few animal lineages. The biosyntheses of polyketides share striking similarities with fatty acid biosynthesis.
Contents
The PKS genes for a certain polyketide are usually organized in one operon in bacteria and in gene clusters in eukaryotes.
Classification
PKSs can be classified into three groups:
Type I PKSs are further subdivided:
Iterative PKSs (IPKSs) can be still further subdivided:
Modules and domains
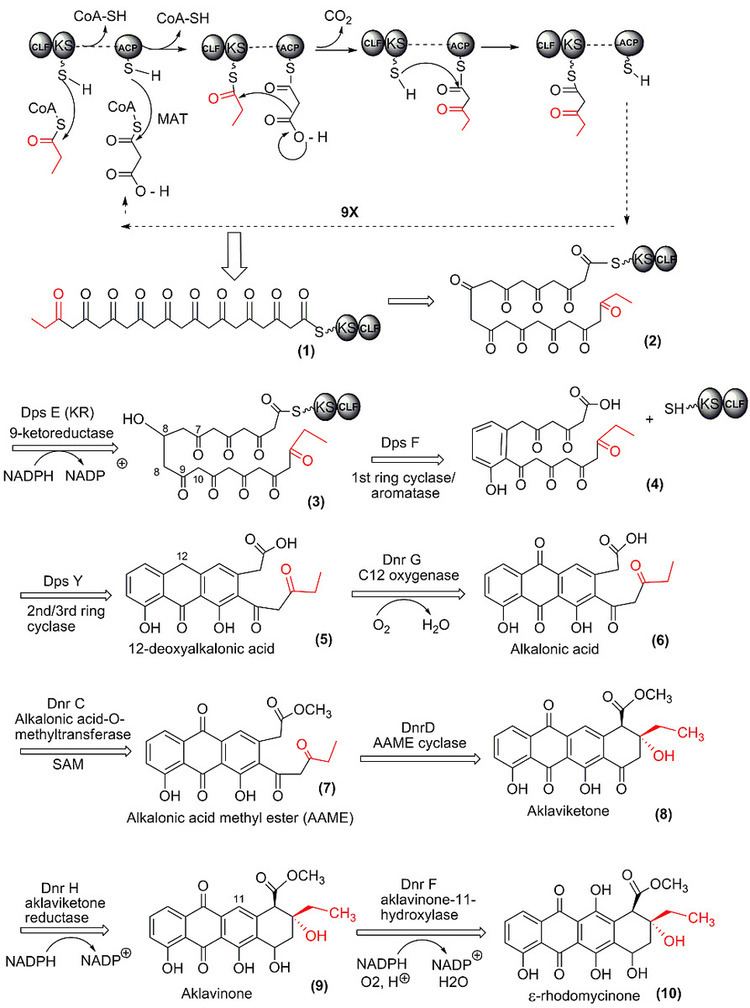
Each type I polyketide-synthase module consists of several domains with defined functions, separated by short spacer regions. The order of modules and domains of a complete polyketide-synthase is as follows (in the order N-terminus to C-terminus):
Domains:
The polyketide chain and the starter groups are bound with their carboxy functional group to the SH groups of the ACP and the KS domain through a thioester linkage: R-C(=O)OH + HS-protein <=> R-C(=O)S-protein + H2O.
Stages
The growing chain is handed over from one thiol group to the next by trans-acylations and is released at the end by hydrolysis or by cyclization (alcoholysis or aminolysis).
Starting stage:
Elongation stages:
Termination stage:
Pharmacological relevance
Polyketide synthases are an important source of naturally occurring small molecules used for chemotherapy. For example, many of the commonly used antibiotics, such as tetracycline and macrolides, are produced by polyketide synthases. Other industrially important polyketides are sirolimus (immunosuppressant), erythromycin (antibiotic), lovastatin (anticholesterol drug), and epothilone B (anticancer drug).
Ecological significance
Only about 1% of all known molecules are natural products, yet it has been recognized that almost two thirds of all drugs currently in use are at least in part derived from a natural source. This bias is commonly explained with the argument that natural products have co-evolved in the environment for long time periods and have therefore been pre-selected for active structures. Polyketide synthase products include lipids with antibiotic, antifungal, antitumor, and predator-defense properties; however, many of the polyketide synthase pathways that bacteria, fungi and plants commonly use have not yet been characterized. Methods for the detection of novel polyketide synthase pathways in the environment have therefore been developed. Molecular evidence supports the notion that many novel polyketides remain to be discovered from bacterial sources.
