Appearance white powder Formula (C6H10O5)n | Related compounds Density 1.5 g/cm³ | |
 | ||
3 structure of cellulose hsc chemistry
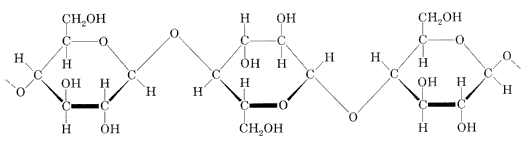
Cellulose is an organic compound with the formula (C
6H
10O
5)
n, a polysaccharide consisting of a linear chain of several hundred to many thousands of β(1→4) linked -glucose units. Cellulose is an important structural component of the primary cell wall of green plants, many forms of algae and the oomycetes. Some species of bacteria secrete it to form biofilms. Cellulose is the most abundant organic polymer on Earth. The cellulose content of cotton fiber is 90%, that of wood is 40–50% and that of dried hemp is approximately 57%.
Contents
- 3 structure of cellulose hsc chemistry
- B 10 starch glycogen and cellulose hl
- History
- Structure and properties
- Assay
- Biosynthesis
- Breakdown cellulolysis
- Breakdown thermolysis
- Hemicellulose
- Derivatives
- Applications
- References
Cellulose is mainly used to produce paperboard and paper. Smaller quantities are converted into a wide variety of derivative products such as cellophane and rayon. Conversion of cellulose from energy crops into biofuels such as cellulosic ethanol is under investigation as an alternative fuel source. Cellulose for industrial use is mainly obtained from wood pulp and cotton.
Some animals, particularly ruminants and termites, can digest cellulose with the help of symbiotic micro-organisms that live in their guts, such as Trichonympha. In humans, cellulose acts as a hydrophilic bulking agent for feces and is often referred to as a "dietary fiber".
B 10 starch glycogen and cellulose hl
History
Cellulose was discovered in 1838 by the French chemist Anselme Payen, who isolated it from plant matter and determined its chemical formula. Cellulose was used to produce the first successful thermoplastic polymer, celluloid, by Hyatt Manufacturing Company in 1870. Production of rayon ("artificial silk") from cellulose began in the 1890s and cellophane was invented in 1912. Hermann Staudinger determined the polymer structure of cellulose in 1920. The compound was first chemically synthesized (without the use of any biologically derived enzymes) in 1992, by Kobayashi and Shoda.
Structure and properties
Cellulose has no taste, is odorless, is hydrophilic with the contact angle of 20–30 degrees, is insoluble in water and most organic solvents, is chiral and is biodegradable. It was shown to melt at 467 °C in 2016. It can be broken down chemically into its glucose units by treating it with concentrated mineral acids at high temperature.
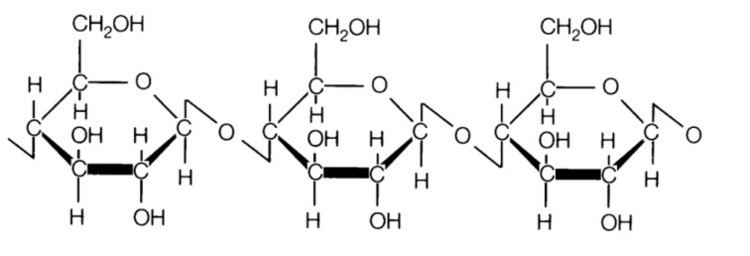
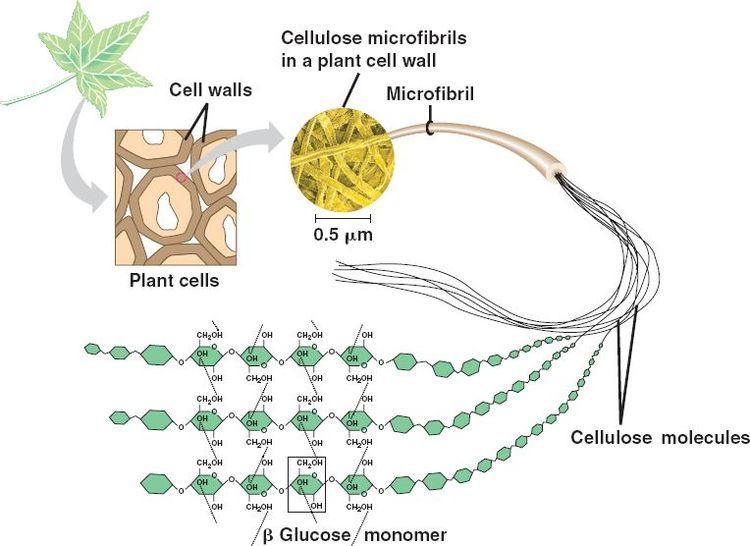
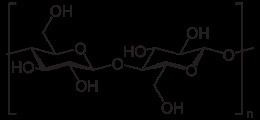
Cellulose is derived from D-glucose units, which condense through β(1→4)-glycosidic bonds. This linkage motif contrasts with that for α(1→4)-glycosidic bonds present in starch and glycogen. Cellulose is a straight chain polymer: unlike starch, no coiling or branching occurs, and the molecule adopts an extended and rather stiff rod-like conformation, aided by the equatorial conformation of the glucose residues. The multiple hydroxyl groups on the glucose from one chain form hydrogen bonds with oxygen atoms on the same or on a neighbor chain, holding the chains firmly together side-by-side and forming microfibrils with high tensile strength. This confers tensile strength in cell walls, where cellulose microfibrils are meshed into a polysaccharide matrix.
Compared to starch, cellulose is also much more crystalline. Whereas starch undergoes a crystalline to amorphous transition when heated beyond 60–70 °C in water (as in cooking), cellulose requires a temperature of 320 °C and pressure of 25 MPa to become amorphous in water.
Several different crystalline structures of cellulose are known, corresponding to the location of hydrogen bonds between and within strands. Natural cellulose is cellulose I, with structures Iα and Iβ. Cellulose produced by bacteria and algae is enriched in Iα while cellulose of higher plants consists mainly of Iβ. Cellulose in regenerated cellulose fibers is cellulose II. The conversion of cellulose I to cellulose II is irreversible, suggesting that cellulose I is metastable and cellulose II is stable. With various chemical treatments it is possible to produce the structures cellulose III and cellulose IV.
Many properties of cellulose depend on its chain length or degree of polymerization, the number of glucose units that make up one polymer molecule. Cellulose from wood pulp has typical chain lengths between 300 and 1700 units; cotton and other plant fibers as well as bacterial cellulose have chain lengths ranging from 800 to 10,000 units. Molecules with very small chain length resulting from the breakdown of cellulose are known as cellodextrins; in contrast to long-chain cellulose, cellodextrins are typically soluble in water and organic solvents.
Plant-derived cellulose is usually found in a mixture with hemicellulose, lignin, pectin and other substances, while bacterial cellulose is quite pure, has a much higher water content and higher tensile strength due to higher chain lengths.
Cellulose is soluble in Schweizer's reagent, cupriethylenediamine (CED), cadmiumethylenediamine (Cadoxen), N-methylmorpholine N-oxide, and lithium chloride / dimethylacetamide. This is used in the production of regenerated celluloses (such as viscose and cellophane) from dissolving pulp. Cellulose is also soluble in many kinds of ionic liquids.
Cellulose consists of crystalline and amorphous regions. By treating it with strong acid, the amorphous regions can be broken up, thereby producing nanocrystalline cellulose, a novel material with many desirable properties. Recently, nanocrystalline cellulose was used as the filler phase in bio-based polymer matrices to produce nanocomposites with superior thermal and mechanical properties.
Assay
Given a cellulose-containing material, the carbohydrate portion that does not dissolve in a 17.5% solution of sodium hydroxide at 20 °C is α cellulose, which is true cellulose. Acidification of the extract precipitates β cellulose. The portion that dissolves in base but does not precipitate with acid is γ cellulose.
Cellulose can be assayed using a method described by Updegraff in 1969, where the fiber is dissolved in acetic and nitric acid to remove lignin, hemicellulose, and xylosans. The resulting cellulose is allowed to react with anthrone in sulfuric acid. The resulting coloured compound is assayed spectrophotometrically at a wavelength of approximately 635 nm.
In addition, cellulose is represented by the difference between acid detergent fiber (ADF) and acid detergent lignin (ADL).
Luminescent conjugated oligothiophenes can also be used to detect cellulose using fluorescence microscopy or spectrofluorometric methods.
Biosynthesis
In vascular plants cellulose is synthesized at the plasma membrane by rosette terminal complexes (RTCs). The RTCs are hexameric protein structures, approximately 25 nm in diameter, that contain the cellulose synthase enzymes that synthesise the individual cellulose chains. Each RTC floats in the cell's plasma membrane and "spins" a microfibril into the cell wall.
RTCs contain at least three different cellulose synthases, encoded by CesA genes, in an unknown stoichiometry. Separate sets of CesA genes are involved in primary and secondary cell wall biosynthesis. There are known to be about seven subfamilies in the CesA superfamily. These cellulose synthases use UDP-glucose to form the β(1→4)-linked cellulose.
Cellulose synthesis requires chain initiation and elongation, and the two processes are separate. CesA glucosyltransferase initiates cellulose polymerization using a steroid primer, sitosterol-beta-glucoside, and UDP-glucose. Cellulose synthase utilizes UDP-D-glucose precursors to elongate the growing cellulose chain. A cellulase may function to cleave the primer from the mature chain.
Cellulose is also synthesised by animals, particularly in the tests of ascidians (where the cellulose was historically termed "tunicine") although it is also a minor component of mammalian connective tissue.
Breakdown (cellulolysis)
Cellulolysis is the process of breaking down cellulose into smaller polysaccharides called cellodextrins or completely into glucose units; this is a hydrolysis reaction. Because cellulose molecules bind strongly to each other, cellulolysis is relatively difficult compared to the breakdown of other polysaccharides. However, this process can be significantly intensified in a proper solvent, e.g. in an ionic liquid.
Most mammals have limited ability to digest dietary fiber such as cellulose. Some ruminants like cows and sheep contain certain symbiotic anaerobic bacteria (like Cellulomonas) in the flora of the rumen, and these bacteria produce enzymes called cellulases that help the microorganism to digest cellulose; the breakdown products are then used by the bacteria for proliferation. The bacterial mass is later digested by the ruminant in its digestive system (stomach and small intestine). Horses use cellulose in their diet by fermentation in their hindgut via symbiotic bacteria which produce cellulase to digest cellulose. Similarly, some termites contain in their hindguts certain flagellate protozoa producing such enzymes, whereas others contain bacteria or may produce cellulase.
The enzymes used to cleave the glycosidic linkage in cellulose are glycoside hydrolases including endo-acting cellulases and exo-acting glucosidases. Such enzymes are usually secreted as part of multienzyme complexes that may include dockerins and carbohydrate-binding modules.
Breakdown (thermolysis)
At temperatures above 350 °C, cellulose undergoes thermolysis (also called ‘pyrolysis’), decomposing into solid char, vapors, aerosols, and gases such as carbon dioxide. Maximum yield of vapors which condense to a liquid called ‘bio-oil’ is obtained at 500 °C.
Semi-crystalline cellulose polymers react at pyrolysis temperatures (350 – 600 °C) in a few seconds; this transformation has been shown to occur via a solid-to-liquid-to-vapor transition, with the liquid (called ‘intermediate liquid cellulose’ or 'molten cellulose') existing for only a fraction of a second. Glycosidic bond cleavage produces short cellulose chains of two-to-seven monomers comprising the melt. Vapor bubbling of intermediate liquid cellulose produces aerosols, which consist of short chain anhydro-oligomers derived from the melt.
Continuing decomposition of molten cellulose produces volatile compounds including levoglucosan, furans, pyrans, light oxygenates and gases via primary reactions. Within thick cellulose samples, volatile compounds such as levoglucosan undergo ‘secondary reactions’ to volatile products including pyrans and light oxygenates such as glycolaldehyde.
Hemicellulose
Hemicellulose is a polysaccharide related to cellulose that comprises about 20% of the biomass of most plants. In contrast to cellulose, hemicellulose is derived from several sugars in addition to glucose, especially xylose but also including mannose, galactose, rhamnose, and arabinose. Hemicellulose consists of shorter chains – between 500 and 3000 sugar units. Furthermore, hemicellulose is branched, whereas cellulose is unbranched.
Derivatives
The hydroxyl groups (-OH) of cellulose can be partially or fully reacted with various reagents to afford derivatives with useful properties like mainly cellulose esters and cellulose ethers (-OR). In principle, though not always in current industrial practice, cellulosic polymers are renewable resources.
Ester derivatives include:
The cellulose acetate and cellulose triacetate are film- and fiber-forming materials that find a variety of uses. The nitrocellulose was initially used as an explosive and was an early film forming material. With camphor, nitrocellulose gives celluloid.
Ether derivatives include:
The sodium carboxymethyl cellulose can be cross-linked to give the croscarmellose sodium (E468) for use as a disintegrant in pharmaceutical formulations.
Applications
Cellulose for industrial use is mainly obtained from wood pulp and cotton. The kraft process is used to separate cellulose from lignin, another major component of plant matter.
