Formula C6H5CH2- | ||
 | ||
The difference between phenyl and benzyl groups
In organic chemistry, benzyl is the substituent or molecular fragment possessing the structure C6H5CH2–. Benzyl features a benzene ring attached to a CH2 group.
Contents
- The difference between phenyl and benzyl groups
- Nomenclature
- Abbreviations
- Reactivity of benzylic centers
- Alcohol protection
- Most common protection methods
- Most common deprotection methods
- The p methoxybenzyl protecting group
- Amine protection
- Most common amine protection methods
- Most common amine deprotection methods
- References
Nomenclature
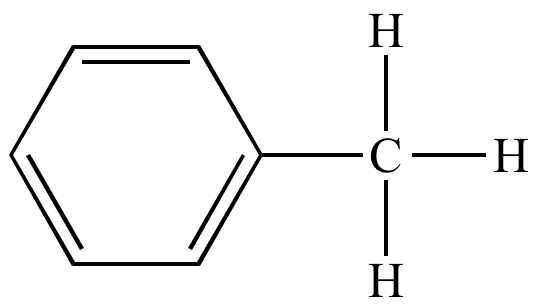
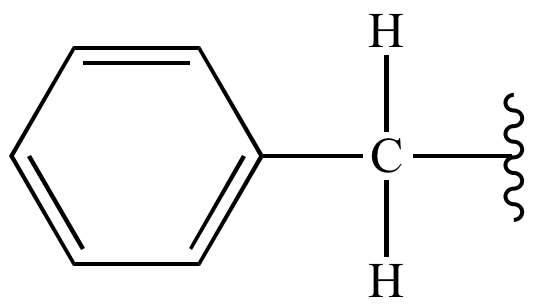
In IUPAC nomenclature the prefix benzyl refers to a C6H5CH2 substituent, for example benzyl chloride or benzyl benzoate. Benzyl is not to be confused with phenyl with the formula C6H5. The term benzylic is used to describe the position of the first carbon bonded to a benzene or other aromatic ring. For example, the molecule, is referred to as a "benzylic" carbocation. The benzyl free radical has the formula C
6H
5CH•
2. The benzylium carbocation has the formula C
6H
5CH+
2; the carbanion has the formula C
6H
5CH−
2. None of these species can be formed in significant amounts under normal conditions, but they are useful referents for discussion of reaction mechanisms.
Abbreviations
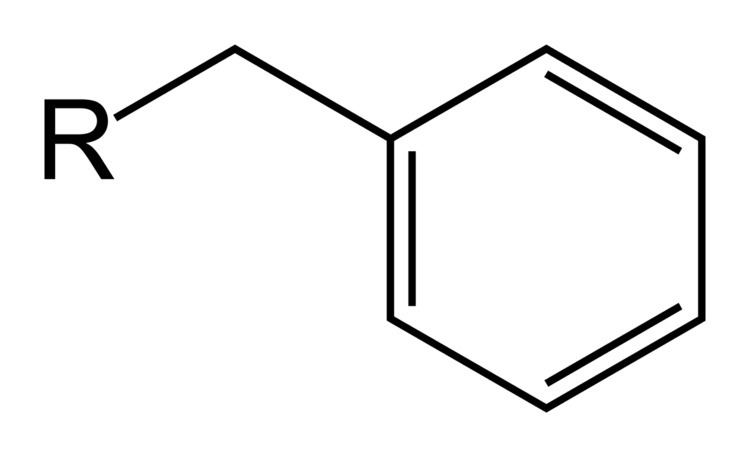
The abbreviation "Bn" is frequently used to denote benzyl groups in nomenclature and structural depictions of chemical compounds. For example, benzyl alcohol can be represented as BnOH. This abbreviation is not to be confused with "Bz", which is the abbreviation for the benzoyl group C6H5C(O)−, or the phenyl group C6H5, abbreviated "Ph".
Reactivity of benzylic centers

The enhanced reactivity of benzylic positions is attributed to the low bond dissociation energy for benzylic C−H bonds. Specifically, the bond C6H5CH2−H is about 10–15% weaker than other kinds of C−H bonds. The neighboring aromatic ring stabilizes benzyl radicals. The data tabulated below compare benzylic C−H bond to related C−H bond strengths.

The weakness of the C−H bond reflects the stability of the benzylic radical. For related reasons, benzylic substituents exhibit enhanced reactivity, as in oxidation, free radical halogenation, or hydrogenolysis. As a practical example, in the presence of suitable catalysts, p-xylene oxidizes exclusively at the benzylic positions to give terephthalic acid:
CH3C6H4CH3 + 3 O2 → HO2CC6H4CO2H + 2 H2O.Millions of tonnes of terephthalic acid are produced annually by this method.
Alcohol protection
Benzyl, abbreviated as Bn, is commonly used in organic synthesis as a robust protecting group for alcohols and carboxylic acids.
Most common protection methods
Most common deprotection methods
Benzyl ethers can be removed under reductive conditions, oxidative conditions, and the use of Lewis Acids.
Reductive conditions
Oxidative conditions
Lewis acid-based
The p-methoxybenzyl protecting group
p-Methoxybenzyl (PMB) is used as a protecting group for alcohols in organic synthesis.
Most common protection methods
Most common deprotection methods
Amine protection
The benzyl group is largely used as a protecting group for amines in organic synthesis.
