Subclass Rickettsidae Family Rickettsiaceae Higher classification Rickettsiaceae | Scientific name Wolbachia | |
 | ||
Similar Bacteria, Rickettsia, Aedes, Yellow fever mosquito, Drosophila simulans | ||
What is wolbachia
Wolbachia is a genus of bacteria which infects arthropod species, including a high proportion of insects, but also some nematodes. It is one of the world's most common parasitic microbes and is possibly the most common reproductive parasite in the biosphere. Its interactions with its hosts are often complex, and in some cases have evolved to be mutualistic rather than parasitic. Some host species cannot reproduce, or even survive, without Wolbachia infection. One study concluded that more than 16% of neotropical insect species carry bacteria of this genus, and as many as 25 to 70% of all insect species are estimated to be potential hosts.
Contents
- What is wolbachia
- Wolbachia the bacteria that could change everything
- History
- Method of sexual differentiation in hosts
- Effects of sexual differentiation in hosts
- Fitness advantages by Wolbachia infections
- Genomics
- Small RNA
- Horizontal gene transfer
- Applications to human related infections
- References
Wolbachia the bacteria that could change everything
History
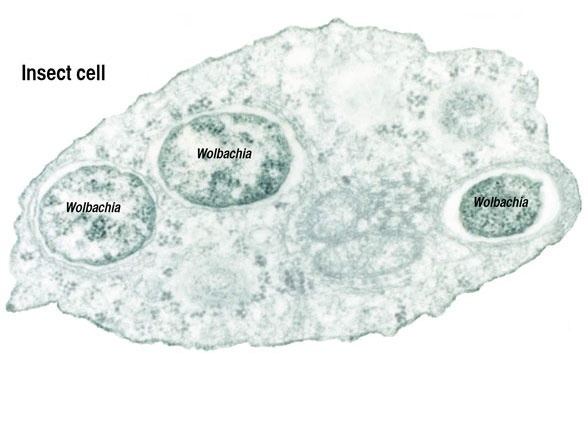
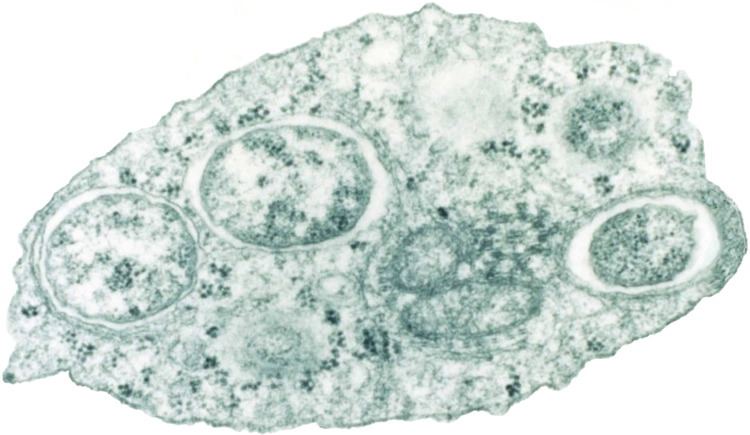
The genus was first identified in 1924 by Marshall Hertig and S. Burt Wolbach in Culex pipiens, the common house mosquito. Hertig formally described the species in 1936 as Wolbachia pipientis. Research on Wolbachia intensified after 1971, when Janice Yen and A. Ralph Barr of UCLA discovered that Culex mosquito eggs were killed by a cytoplasmic incompatibility when the sperm of Wolbachia-infected males fertilized infection-free eggs. The genus Wolbachia is of considerable interest today due to its ubiquitous distribution, its many different evolutionary interactions, and its potential use as a biocontrol agent.
Method of sexual differentiation in hosts
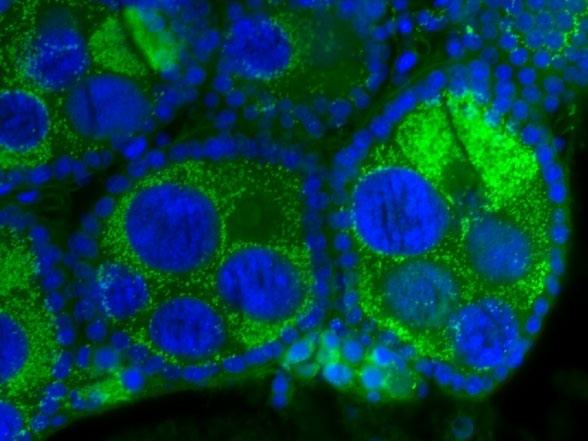
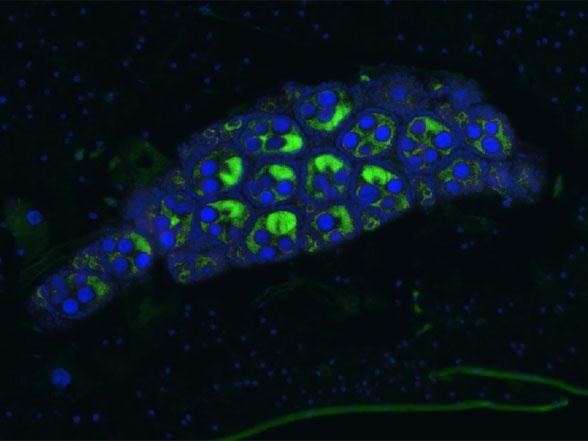
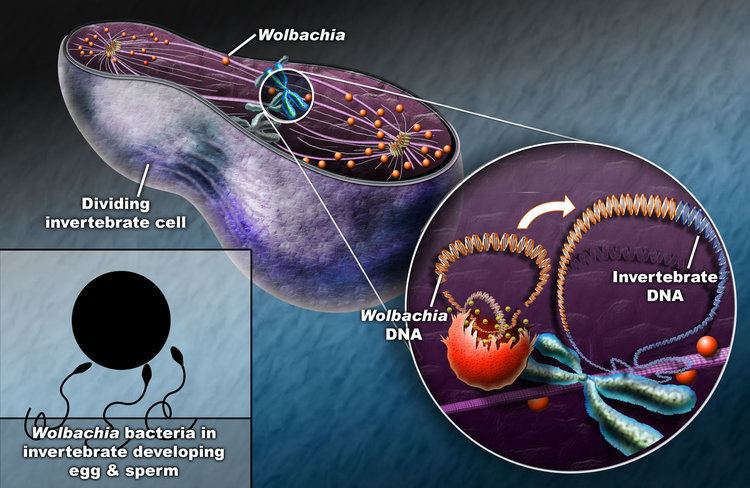
These bacteria can infect many different types of organs, but are most notable for the infections of the testes and ovaries of their hosts. Wolbachia species are ubiquitous in mature eggs, but not mature sperm. Only infected females, therefore, pass the infection on to their offspring. Wolbachia bacteria maximize their spread by significantly altering the reproductive capabilities of their hosts, with four different phenotypes:
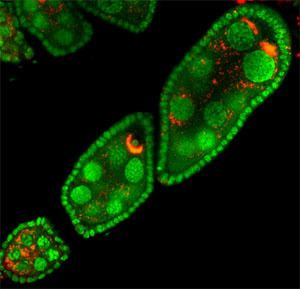
Effects of sexual differentiation in hosts
Several host species, such as Trichogramma, are so dependent on sexual differentiation of Wolbachia that they are unable to reproduce effectively without the bacteria in their bodies, and some might even be unable to survive uninfected.
One study on infected woodlice showed the broods of infected organisms had a higher proportion of females than their uninfected counterparts.
Wolbachia, especially Wolbachia-caused cytoplasmic incompatibility, may be important in promoting speciation. Wolbachia strains that distort the sex ratio may alter their host's pattern of sexual selection in nature, and also engender strong selection to prevent their action, leading to some of the fastest examples of natural selection in natural populations.
The male killing and feminization effects of Wolbachia infections can also lead to speciation in their hosts. For example, populations of the pill woodlouse, Armadillidium vulgare, which are exposed to the feminizing effects of Wolbachia, have been known to lose their female-determining chromosome. In these cases, only the presence of Wolbachia can cause an individual to develop into a female.
Fitness advantages by Wolbachia infections
Wolbachia has been linked to viral resistance in Drosophila melanogaster, Drosophila simulans, and mosquito species. Flies infected with the bacteria are more resistant to RNA viruses such as Drosophila C virus, Nora virus, flock house virus, cricket paralysis virus, chikungunya virus, and West Nile virus.
In the common house mosquito, higher levels of Wolbachia were correlated with more insecticide resistance.
In leafminers of the species Phyllonorycter blancardella, Wolbachia bacteria help their hosts produce green islands on yellowing tree leaves, that is, small areas of leaf remaining fresh, allowing the hosts to continue feeding while growing to their adult forms. Larvae treated with tetracycline, which kills Wolbachia, lose this ability and subsequently only 13% emerge successfully as adult moths.
The Muscidifurax uniraptor parasitoid wasp also benefits from hosting a Wolbachia bacterium. Faculty from the Hebrew University of Jerusalem studied the positive effects of Wolbachia on the fecundity of a strain of M. uniraptor from Rochester, New York, in 1997.
In the parasitic filarial nematode species responsible for elephantiasis, such as Brugia malayi and Wuchereria bancrofti, Wolbachia has become an obligate endosymbiont and provides the host with chemicals necessary to its reproduction and survival. Elimination of the Wolbachia symbionts through antibiotic treatment therefore prevents reproduction of the nematode, and eventually results in its premature death.
Some Wolbachia species that infect arthropods also provide some metabolic provisioning to their hosts. In Drosophila melanogaster, Wolbachia is found to mediate iron metabolism under nutritional stress and in Cimex lectularis, Wolbachia strain cCle helps the host to synthesize B vitamins.
Wolbachia is also found to provide the host with a benefit of increasing fecundity. Wolbachia strains captured from 1988 in southern California still induce a fecundity deficit, but nowadays the fecundity deficit is replaced with a fecundity advantage such that infected Drosophila simulans produces more offspring than the uninfected ones.
Genomics
The first Wolbachia genome to be determined was that of one that infects D. melanogaster fruit flies. This genome was sequenced at The Institute for Genomic Research in a collaboration between Jonathan Eisen and Scott O'Neill. The second Wolbachia genome to be determined was one that infects Brugia malayi nematodes. Genome sequencing projects for several other Wolbachia strains are in progress. A nearly complete copy of the Wolbachia genome sequence was found within the genome sequence of the fruit fly Drosophila ananassae and large segments were found in seven other Drosophila species.
In an application of DNA barcoding to the identification of species of Protocalliphora flies, several distinct morphospecies had identical cytochrome c oxidase I gene sequences, most likely through horizontal gene transfer by Wolbachia species as they jump across host species. As a result, Wolbachia can cause misleading results in molecular cladistical analyses.
Small RNA
Wolbachia's small non-coding RNAs WsnRNA-46 and WsnRNA-59 were detected in Aedes aegypti mosquitoes and Drosophila melanogaster. The small RNAs may regulate bacterial and host genes.
Horizontal gene transfer
Wolbachia species also harbor a temperate bacteriophage called WO. Comparative sequence analyses of bacteriophage WO offer some of the most compelling examples of large-scale horizontal gene transfer between Wolbachia coinfections in the same host. It is the first bacteriophage implicated in frequent lateral transfer between the genomes of bacterial endosymbionts. Gene transfer by bacteriophages could drive significant evolutionary change in the genomes of intracellular bacteria that were previously considered highly stable or prone to loss of genes over time.
Applications to human-related infections
Outside of insects, Wolbachia infects a variety of isopod species, spiders, mites, and many species of filarial nematodes (a type of parasitic worm), including those causing onchocerciasis (river blindness) and elephantiasis in humans, as well as heartworms in dogs. Not only are these disease-causing filarial worms infected with Wolbachia, but Wolbachia also seems to play an inordinate role in these diseases. A large part of the pathogenicity of filarial nematodes is due to host immune response toward their Wolbachia. Elimination of Wolbachia from filarial nematodes generally results in either death or sterility of the nematode. Consequently, current strategies for control of filarial nematode diseases include elimination of their symbiotic Wolbachia via the simple doxycycline antibiotic, rather than directly killing the nematode with far more toxic antinematode medications.
Naturally existing strains of Wolbachia have been shown to be a route for vector control strategies because of their presence in arthropod populations, such as mosquitos. Due to the unique traits of Wolbachia that cause cytoplasmic incompatibility, this strain is useful as a promoter of genetic drive within a population. Wolbachia-infected females are able to produce offspring with uninfected and infected males, however, uninfected females are only able to produce viable offspring with uninfected males. This gives infected females a frequency-dependent reproductive advantage; that is, the advantage is greater the higher the frequency of Wolbachia in the population. Computational models predict that introducing Wolbachia strains into natural populations will reduce pathogen transmission and reduce overall disease burden. An example includes Wolbachia that can be used to control dengue and malaria by eliminating the older insects that contain more parasites. Promoting the survival and reproduction of younger insects lessens selection pressure for evolution of resistance. Wolbachia strains that are able to reduce dengue transmission include wAllbB and wMelPop with Aedes aegypti, wMel with Aedes albopictus. and Aedes aegypti. In addition to inhibit the dengue virus (DENV) in some species of genus Aedes, Wolbachia has been identified to inhibit replication of chikungunya virus (CHIKV) in A. aegypti. The Wmel strain of Wolbachia pipientis significantly reduced infection and dissemination rates of CHIKV in mosquitoes, compared to Wolbachia uninfected controls and the same phenomenon was observed in yellow fever virus infection converting this bacterium in an excellent promise for YFV and CHIKV suppression.
Another study tested the effect of Wolbachia on the replication of West Nile virus (WNV), but in cell line Aag2 derived from A. aegypti cells, suggesting that despite the existence of an enhancement of viral genomic RNA replication in the cell line Wolbachia infected, the production of secreted virus was significantly inhibited. Also, the antiviral effect in intrathoracically infected mosquitoes depends on the strain of Wolbachia, and the replication of the virus in orally fed mosquitoes was completely inhibited in wMelPop strain of Wolbachia.
However, cases where Wolbachia reduces transmission of pathogens must be studied carefully. Indeed, a recent study demonstrated that the West Nile virus could be improved by the presence of the Wolbachia strain wAlbB in the mosquito Culex tarsalis. The rate of virus appears significantly higher than in uninfected mosquitoes. Indeed, wAlbB inhibits REL1, which is an activator of the antiviral Toll immune pathway. The release of artificially infected mosquitoes in the environment, for a vector-borne disease control program, should be made with caution, because it is the first case of improvement of a human disease in the mosquito.
Wolbachia may induce reactive oxygen species-dependent activation of the Toll (gene family) pathway, which is essential for activation of antimicrobial peptides, defensins, and cecropins that help to inhibit dengue virus proliferation. Wolbachia infection can also increase mosquito resistance to malaria, as shown in Anopheles stephensi where the wAlbB strain of Wolbachia hindered the lifecycle of Plasmodium falciparum.
Some experts have proposed to combat the spread of the Zika virus by breeding and releasing mosquitoes that have intentionally been infected with an appropriate strain of Wolbachia. A recent study has shown that Wolbachia has the ability to block the spread of Zika virus in mosquitos in Brazil.
In October 2016, it was announced that $18 million in funding was being allocated for the use of Wolbachia-infected mosquitoes to fight Zika and dengue viruses. Deployment is slated for early 2017 in Colombia and Brazil.
