Genus Wuchereria Rank Species | Suborder Spirurina Scientific name Wuchereria bancrofti | |
 | ||
Similar Filarioidea, Culex, Tropical eosinophilia | ||
Parasitology wuchereria bancrofti
Wuchereria bancrofti is a human parasitic roundworm that is the major cause of lymphatic filariasis. It is one of the three parasitic worms, together with Brugia malayi and B. timori, that infect the lymphatic system to cause Lymphatic filariasis. These filarial worms are spread by a variety of mosquito vector species. W. bancrofti is the most prevalent of the three and affects over 120 million people, primarily in Central Africa and the Nile delta, South and Central America, the tropical regions of Asia including southern China, and the Pacific islands. If left untreated, the infection can develop into a chronic disease called elephantiasis. In rare conditions it also causes tropical eosinophilia, an asthmatic disease. Limited treatment modalities exist and no vaccines have been developed.
Contents
- Parasitology wuchereria bancrofti
- Wuchereria bancrofti life cycle
- History
- Morphology
- Life cycle
- Epidemiology
- Pathology
- Diagnosis
- Treatment
- Control
- Eradication
- References
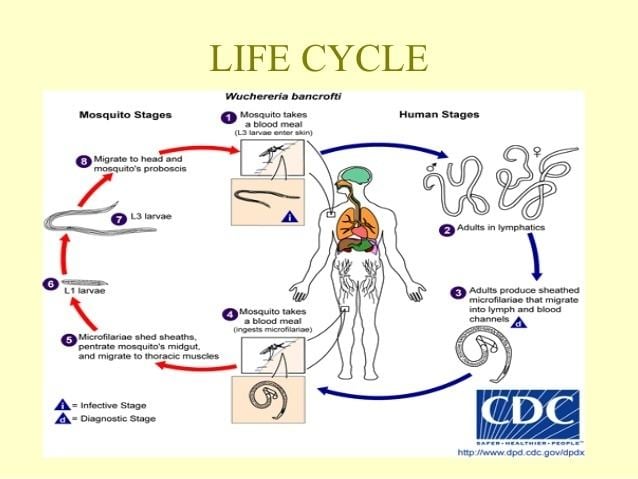
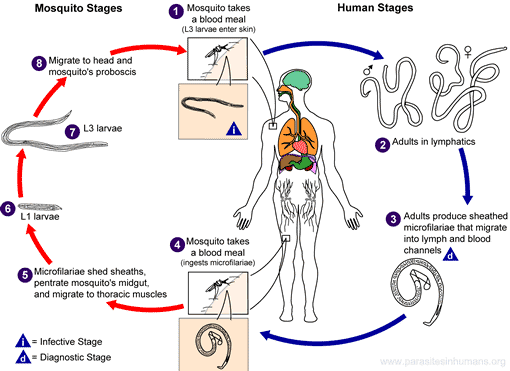
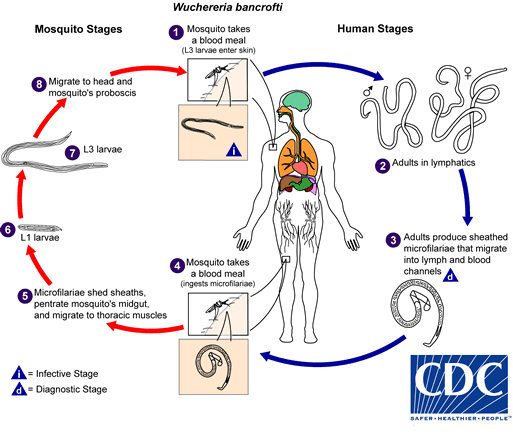
Wuchereria bancrofti life cycle
History
The effects of W. bancrofti were documented early in ancient text. Ancient Greek and Roman writers noted the similarities between the enlarged limbs and cracked skin of infected individuals to that of elephants. Since then, this condition has been commonly known as elephantiasis. However, this is a misnomer, since elephantiasis literally translates to “a disease caused by elephants”.
W. bancrofti was named after physician Otto Wucherer and parasitologist Joseph Bancroft, both of whom extensively studied filarial infections.
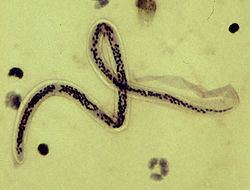
W. bancrofti is speculated to have been brought to the New World by the slave trade. Once it was introduced to the New World, this filarial worm disease persisted throughout the areas surrounding Charleston, South Carolina until its sudden disappearance in the 1920s.
Morphology
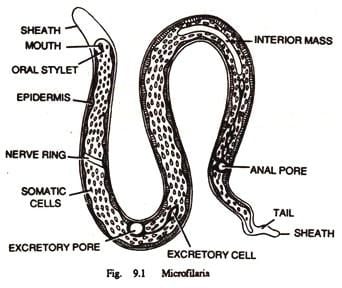
As a dioecious worm, W. bancrofti exhibits sexual dimorphism. The adult worm is long, cylindrical, slender, and smooth with rounded ends. It is white in colour and almost transparent. The body is quite delicate making it difficult to remove from tissues. It has a short cephalic or head region connected to the main body by a short neck which appears as a constriction. There are dark spots which are dispersed nuclei throughout the body cavity, with no nuclei at the tail tip. Male and female can be differentiated by size and structure of tail tip. The male worm is smaller, 40 millimetres (1.6 in) long and 100 micrometres (0.0039 in) wide, and features a ventrally curved tail. The tip of the tail has 15 pairs of minute caudal papillae, the sensory organs. The anal region is an elaborate structure consisting of 12 pairs of papillae, of which 8 are in front and 4 are behind the anus. In contrast, the female is 60 millimetres (2.4 in) to 100 millimetres (3.9 in) long and 300 micrometres (0.012 in) wide, nearly three times larger in diameter than the male. Its tail gradually tapers and rounded at the tip. There are no additional sensory structures. Its vulva lies towards the anterior region, about 0.25 mm from the head. Adult male and female are most often coiled together and are difficult to separate. Females are ovoviviparous and can produce thousands of juveniles known as microfilariae.
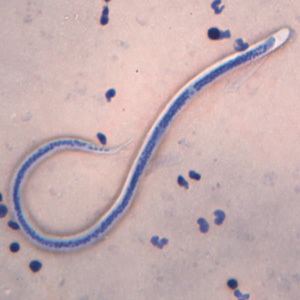
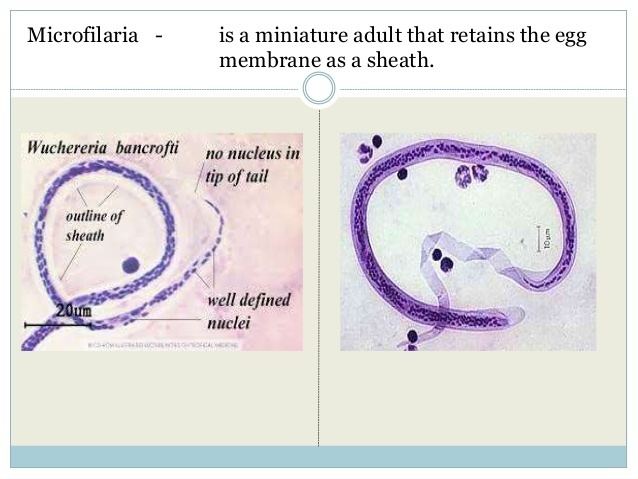
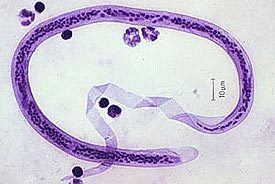
The microfilaria is a miniature adult and retains the egg membrane as a sheath, and is often considered an advanced embryo. It measures 280 μm long and 25 μm wide. It appears quite structureless 'in vivo', but histological staining makes its primitive gut, nerve ring and muscles apparent.
Life cycle
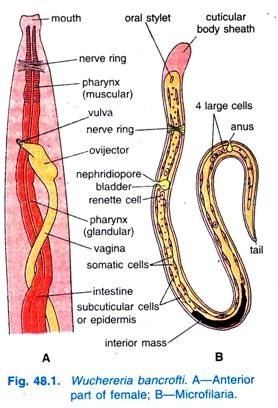
W. bancrofti carry out their life cycle in two hosts. Human beings serve as the definitive host and mosquitoes as their intermediate hosts. The adult parasites reside in the lymphatics of the human host. They are found mostly in the afferent lymphatic channels of the lymph glands in the lower part of the body. The first-stage larvae, known as microfilariae, are present in the circulation. The microfilariae have a membrane "sheath". This sheath, along with the area in which the worms reside, makes identification of the species of microfilariae in Humans easier to determine. The microfilariae are found mainly in the peripheral blood and can be found at peak amounts from 10 p.m. to 4 a.m. They migrate between the deep and the peripheral circulation exhibiting unique diurnal periodicity. During the day, they are present in the deep veins, and during the night, they migrate to the peripheral circulation. The cause of this periodicity remains unknown, but the advantages of the microfilariae being in the peripheral blood during these hours may ensure the vector, the nighttime mosquito, will have a higher chance of transmitting them elsewhere. Physiological changes also are associated with sleeping, such as lowered body temperature, oxygen tension and adrenal activity, and an increased carbon dioxide tension, among other physical alterations, any of which could be the signals for the rhythmic behavior of microfilarial parasites. If the hosts sleep by day and are awake at night, their periodicity is reversed. In the South Pacific, where W. bancrofti shows diurnal periodicity, it is known as periodic.
The microfilariae are transferred into a vector, which are most commonly mosquito species of the genera Culex, Anopheles, Mansonia, and Aedes. Inside the mosquito, the microfilariae mature into motile larvae called juveniles. When the infected mosquito has its next blood meal, W. bancrofti is egested via the mosquito’s proboscis into the blood stream of the new human host. The larvae move through the lymphatic system to regional lymph nodes, predominantly in the legs and genital area. The larvae develop into adult worms over the course of a year, and reach sexual maturity in the afferent lymphatic vessels. After mating, the adult female worm can produce thousands of microfilariae that migrate into the bloodstream. A mosquito vector can bite the infected human host, ingest the microfilariae, and thus repeat the life cycle.
Epidemiology
W. bancrofti is responsible for 90% of lymphatic filariasis. Recently, 120 million worldwide cases of lymphatic filariasis were estimated. W. bancrofti largely affects areas across the broad equatorial belt (Africa, the Nile Delta, Turkey, India, the East Indies, Southeast Asia, Philippines, Oceanic Islands, and parts of South America.)
The mosquito vectors of W. bancrofti have a preference for human blood; humans are apparently the only animals naturally infected with W. bancrofti. There is no reservoir host, and the disease could therefore potentially be eradicated.
Pathology
The pathogenesis of W. bancrofti infection is dependent on the immune system and inflammatory responses of the host. After infection, the worms will mature within 6–8 months, male and female worms will mate and then release the microfilariae. These microfilariae can be released for up to ten years.
- The asymptomatic phase usually consists of high microfilaremia infection, and individuals show no symptoms of being infected. This occurs due to cytokine IL-4 suppressing the activity of TH1 cells in the immune system. This can occur for years until the inflammatory reaction rises again.
- In the inflammatory (acute) phase, the antigens from the female adult worms elicit inflammatory responses. The worms in the lymph channels disrupt the flow of the lymph, causing lymphedema. The individual will exhibit fever, chills, skin infections, painful lymph nodes, and tender skin of the lymphedematous extremity. These symptoms often lessen after five to seven days. Other symptoms that may occur include orchitis, an inflammation of the testes, which is accompanied by painful, immediate enlargement and epididymitis (inflammation of the spermatic cord).
- The obstructive (chronic) phase is marked by lymph varices, lymph scrotum, hydrocele, chyluria (lymph in urine), and elephantiasis. Microfilariae are not normally present in this phase. A key feature of this phase is scar formation from affected tissue areas. Other features include thickening of the skin and elephantiasis, which develops gradually with the attack of the lymphatic system. Elephantiasis affects men mainly in the legs, arms, and scrotum. In women, the legs, arms, and breasts are affected.
Diagnosis
A blood smear is a simple and fairly accurate diagnostic tool, provided the blood sample is taken during the period in the day when the juveniles are in the peripheral circulation. Technicians analyzing the blood smear must be able to distinguish between W. bancrofti and other parasites potentially present.
A polymerase chain reaction test can also be performed to detect a minute fraction, as little as 1 pg, of filarial DNA.
Some infected people do not have microfilariae in their blood. As a result, tests aimed to detect antigens from adult worms can be used.
Ultrasonography can also be used to detect the movements and noises caused by the movement of adult worms.
Dead, calcified worms can be detected by X-ray examinations.
Treatment
The severe symptoms caused by the parasite can be avoided by cleansing the skin, surgery, or the use of anthelmintic drugs, such as diethylcarbamazine (DEC), ivermectin, or albendazole. The drug of choice is DEC, which can eliminate the microfilariae from the blood and also kill the adult worms with a dosage of 6 mg/kg semiannually or annually. A polytherapy treatment that includes ivermectin with DEC or albendazole is more effective than each drug alone. Protection is similar to that of other mosquito-spread illnesses; one can use barriers both physical (a mosquito net), chemical (insect repellent), or mass chemotherapy as a method to control the spread of the disease.
Mass chemotherapy should cover the entire endemic area at the same time. This will significantly decrease the overall microfilarial titer in blood in mass, hence decreasing the transmission through mosquitoes during their subsequent bites.
Antibiotic active against the Wolbachia symbionts of the worm have been experimented with as treatment. Wolbachia-free worms first become sterile, and later die prematurely.
Control
Prevention focuses on protecting against mosquito bites in endemic regions. Insect repellents and mosquito nets are useful to protect against mosquito bites. Public education efforts must also be made within the endemic areas of the world to successfully lower the prevalence of W. bancrofti infections.
Eradication
The WHO is coordinating an effort to eradicate filarisis. The mainstay of this programme is the mass use of antifilarial drugs on a regular basis for at least five years.
In April 2011, Sri Lanka was certified by the WHO as having eradicated this disease.
