3DMet B00133 Formula C3H9N Density 670 kg/m³ | Appearance Colorless gas Boiling point 2.9 °C Molar mass 59.11 g/mol | |
 | ||
Related amines Related compounds | ||
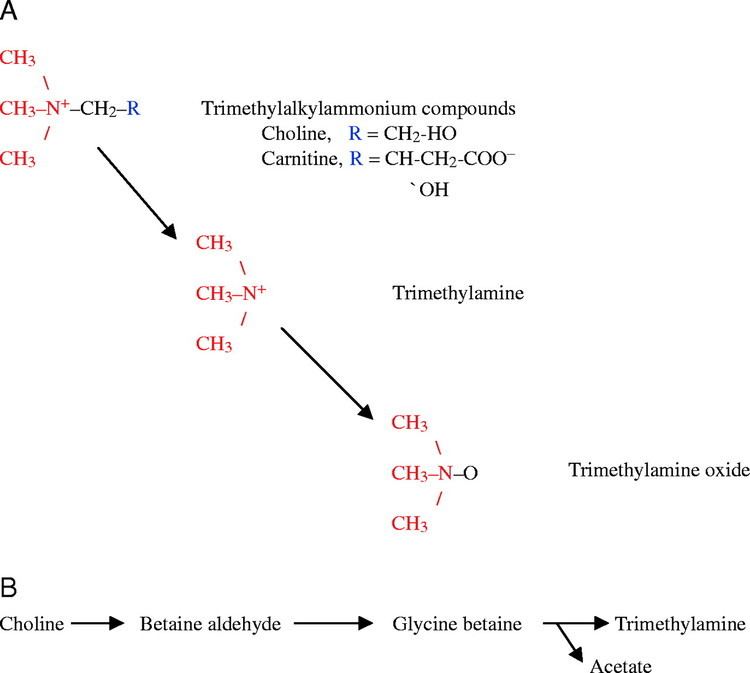
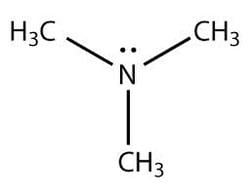
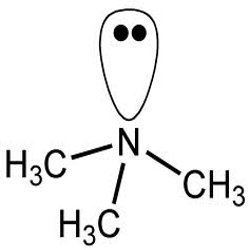
Trimethylamine (TMA) is an organic compound with the formula N(CH3)3. This colorless, hygroscopic, and flammable tertiary amine has a strong "fishy" odor in low concentrations and an ammonia-like odor at higher concentrations. It is a gas at room temperature but is usually sold in pressurized gas cylinders or as a 40% solution in water. TMA is a nitrogenous base and can be readily protonated to give trimethylammonium cation. Trimethylammonium chloride is a hygroscopic colorless solid prepared from hydrochloric acid. Trimethylamine is a good nucleophile, and this reaction is the basis of most of its applications.
Contents
Trimethylamine is a product of decomposition of plants and animals. In humans, it is synthesized exclusively by gut microbiota from dietary nutrients such as choline and carnitine. High levels of trimethylamine are associated with the development of fish odor syndrome, which arise from the foul, fishy odor of trimethylamine. TMA is the substance mainly responsible for the odor often associated with rotting fish, some infections, bad breath and can be a cause of vaginal odor due to bacterial vaginosis. It is also associated with taking large doses of choline and carnitine.

In 2013, trimethylamine was identified as a potent full agonist of human TAAR5, a trace amine-associated receptor that is expressed in the olfactory epithelium and functions as an olfactory receptor for tertiary amines. One or more additional odorant receptors appear to be involved in trimethylamine olfaction in humans as well.
Making trimethylamine stench of rotting fish
Production
Trimethylamine is prepared by the reaction of ammonia and methanol employing a catalyst:
3 CH3OH + NH3 → (CH3)3N + 3 H2OThis reaction coproduces the other methylamines, dimethylamine (CH3)2NH and methylamine CH3NH2.
Trimethylamine has also been prepared via a reaction of ammonium chloride and paraformaldehyde, according to the following equation:
9 (CH2=O)n + 2n NH4Cl → 2n (CH3)3N•HCl + 3n H2O + 3n CO2↑Applications
Trimethylamine is used in the synthesis of choline, tetramethylammonium hydroxide, plant growth regulators or herbicides, strongly basic anion exchange resins, dye leveling agents and a number of basic dyes. Gas sensors to test for fish freshness detect trimethylamine.
Trimethylaminuria
Trimethylaminuria is an autosomal recessive genetic disorder involving a defect in the function or expression of flavin-containing monooxygenase 3 (FMO3) which results in poor trimethylamine metabolism. Individuals with trimethylaminuria develop a characteristic fish odor - the smell of trimethylamine - in their sweat, urine, and breath after the consumption of choline-rich foods. A condition similar to trimethylaminuria has also been observed in a certain breed of Rhode Island Red chicken that produces eggs with a fishy smell, especially after eating food containing a high proportion of rapeseed.