 | ||
Appearance white crystalline solid | ||
Paraformaldehyde meaning
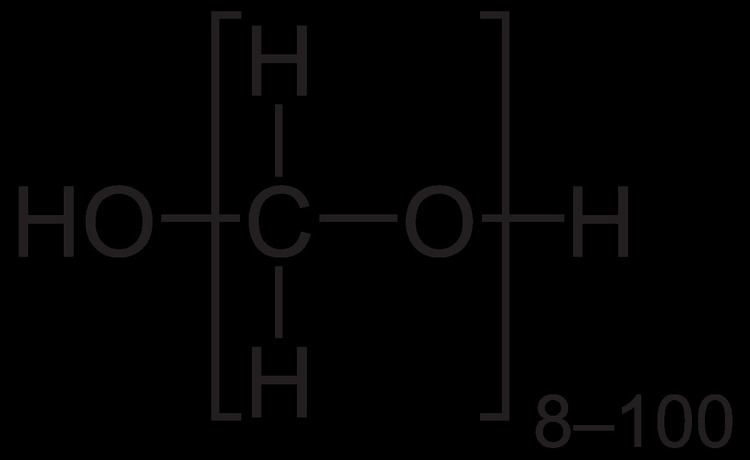
Paraformaldehyde (PFA) is the smallest polyoxymethylene, the polymerization product of formaldehyde with a typical degree of polymerization of 8–100 units. Paraformaldehyde commonly has a slight odor of formaldehyde due to decomposition. Paraformaldehyde is a poly-acetal.
Contents
- Paraformaldehyde meaning
- How to say paraformaldehyde high quality voices
- Synthesis
- Reactions
- Uses
- Toxicity
- References
How to say paraformaldehyde high quality voices
Synthesis
Paraformaldehyde forms slowly in aqueous formaldehyde solutions as a white precipitate, especially if stored in the cold. Formalin actually contains very little monomeric formaldehyde; most of it forms short chains of polyformaldehyde. A small amount of methanol is often added as a stabilizer to limit the extent of polymerization.
Reactions
Paraformaldehyde can be depolymerized to formaldehyde gas by dry heating and to form a formaldehyde solution by water in the presence of a base or heat. The very pure formaldehyde solutions obtained in this way are used as a fixative for microscopy and histology.
The resulting formaldehyde gas from dry heating paraformaldehyde is flammable.
Uses
Once paraformaldehyde is depolymerized, the resulting formaldehyde may be used as a fumigant, disinfectant, fungicide, and fixative. Longer chain-length (high molecular weight) polyoxymethylenes are used as a thermoplastic and are known as polyoxymethylene plastic (POM, Delrin). It was used in the past in the discredited Sargenti method of root canal treatment.
Paraformaldehyde is not a fixative; it must be depolymerized to formaldehyde in solution. In cell culture, a typical formaldehyde fixing procedure would involve using a 4% formaldehyde solution in phosphate buffered saline (PBS) on ice for 10 minutes.
Toxicity
As a formaldehyde releasing agent, paraformaldehyde is a potential carcinogen. Its acute oral median lethal dose in rats is 592 mg/kg.
