 | ||
Chapter 17 oxidation of phenols to quinone
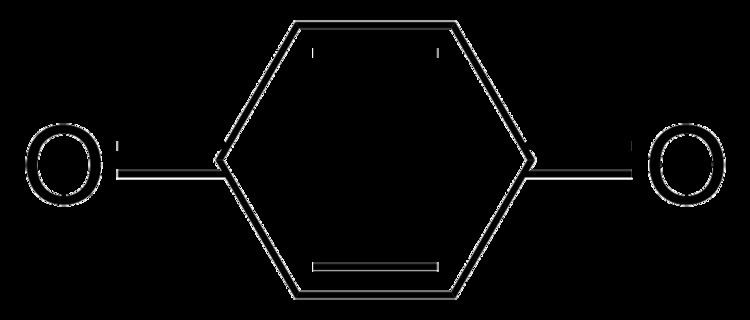
The quinones represent a class of organic compounds that are formally "derived from aromatic compounds [such as benzene or naphthalene] by conversion of an even number of –CH= groups into –C(=O)– groups with any necessary rearrangement of double bonds", resulting in "a fully conjugated cyclic dione structure". The class includes some heterocyclic compounds.
Contents
- Chapter 17 oxidation of phenols to quinone
- Oxidation of dihydoxybenzoquinones to quinones reversal of sulfonation
- Properties
- Production of hydrogen peroxide
- Biochemistry
- Medicinal
- Dyes
- Reagents in organic chemistry
- Battery charge carrier
- Nomenclature
- References
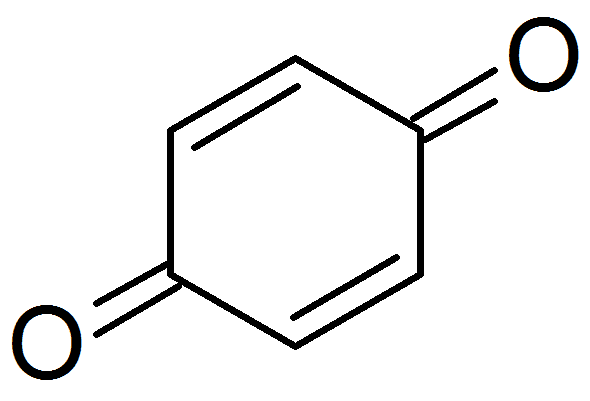
The prototypical member of the class is 1,4-benzoquinone or cyclohexadienedione, often called simply 'quinone' (thus the name of the class). Other important examples are 1,2-benzoquinone (ortho-quinone), 1,4-naphthoquinone and 9,10-anthraquinone.
Oxidation of dihydoxybenzoquinones to quinones reversal of sulfonation
Properties
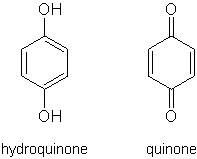
Quinones are oxidized derivatives of aromatic compounds and are often readily made from reactive aromatic compounds with electron-donating substituents such as phenols and catechols, which increase the nucleophilicity of the ring and contributes to the large redox potential needed to break aromaticity. (Quinones are conjugated but not aromatic). Quinones are electrophilic Michael acceptors stabilised by conjugation. Depending on the quinone and the site of reduction, reduction can either rearomatise the compound or break the conjugation. Conjugate addition nearly always breaks the conjugation.
The term quinone is also used more generally for a large class of compounds formally derived from aromatic quinones through replacement of some hydrogen atoms by other atoms or radicals.
Production of hydrogen peroxide
A large scale industrial application of quinones is for the production of hydrogen peroxide. 2-Alkylanthraquinones are hydrogenated to the corresponding hydroquinones (quinizarins), which then transfer H
2 to oxygen:
2 → anthraquinone + H
2O
2
In this way, several billion kilograms of H
2O
2 are produced annually.
Biochemistry
Derivatives of quinones are common in biologically active molecules. Some serve as electron acceptors in electron transport chains such as those in photosynthesis (plastoquinone, phylloquinone), and aerobic respiration (ubiquinone). Phylloquinone is also known as Vitamin K1 as it is used by animals to help form certain proteins, which are involved in blood coagulation, bone formation, and other processes.
A natural example of the oxidation of hydroquinone to quinone is in the spray of bombardier beetles; hydroquinone is reacted with hydrogen peroxide to produce a fiery blast of steam, a strong deterrent in the animal world.
Medicinal
Natural or synthetic quinones show a biological or pharmacological activity, and some of them show anti-tumoral activity. They embody some claims in herbal medicine. These applications include purgative (sennosides), antimicrobial and antiparasitic (rhein- and saprorthoquinone, atovaquone), anti-tumor (emodin and juglone), inhibition of PGE2 biosynthesis (arnebinone and arnebifuranone) and anti-cardiovascular disease (tanshinone).
Dyes
Many natural and artificial coloring substances (dyes and pigments) are quinone derivatives. They are second only to azo dyes in importance as dyestuffs, with particular emphasis on blue colors. Alizarin (1,2-dihydroxy-9,10-anthraquinone), extracted from the madder plant, was the first natural dye to be synthesized from coal tar.
Reagents in organic chemistry
Benzoquinone is used in organic chemistry as an oxidizing agent. Strongly oxidizing quinones include chloranil and 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (also known as DDQ).
Battery charge carrier
9,10-Anthraquinone-2,7-disulphonic acid (AQDS) a quinone similar to one found naturally in rhubarb has been used as a charge carrier in metal-free flow batteries.
Nomenclature
Quinones are commonly named with a prefix that indicates the parent aromatic hydrocarbon ("benzo-" for benzene, "naphtho-" for naphthalene, "anthra-" for anthracene, etc.) and the "-quinone" suffix. Infix multipliers "-di-", "-tri-", "-tetra-" (etc.) are used when there are 4, 6, 8 (etc.) carbonyls. The position of the carbonyl groups can be indicated before the prefix (as in "1,4,5,8-naphthodiquinone") or after it ("anthra-1,4-quinone").
