Abbreviations DDQ Density 1.7 g/cm³ Appearance yellow to orange powder | Formula C8Cl2N2O2 Boiling point 301.8 °C | |
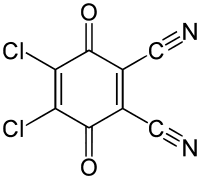 | ||
2,3-Dichloro-5,6-dicyano-1,4-benzoquinone (or DDQ) is the chemical reagent with formula C6Cl2(CN)2O2. This oxidant is useful for the dehydrogenation of alcohols, phenols, and steroid ketones in organic chemistry. DDQ decomposes in water, but is stable in aqueous mineral acid.
Contents
Preparation
Synthesis of DDQ involves cyanation of chloranil. Thiele and Günther first reported a 6-step preparation in 1906. The substance did not receive interest until its potential as a dehydrogenation agent was discovered. A single-step chlorination from 2,3-dicyanohydroquinone was reported in 1965.
Reactions
The reagent removes pairs of H atoms from organic molecules. The stoichiometry of its action is illustrated by the conversion of tetralin to naphthalene:
2 C6Cl2(CN)2O2 + C10H12 → 2 C6Cl2(CN)2(OH)2 + C10H8The resulting hydroquinone is poorly soluble in typical reaction solvents (dioxane, alkanes), which facilitates workup.
Safety
DDQ reacts with water with release of hydrogen cyanide (HCN), which is highly toxic. A low-temperature and weakly acidic environment increases the stability of DDQ.
