Entrez 5660 | Ensembl ENSG00000197746 | |
 | ||
Aliases PSAP, GLBA, SAP1, prosaposin External IDs OMIM: 176801 MGI: 97783 HomoloGene: 37680 GeneCards: PSAP | ||
Prosaposin, also known as PSAP, is a protein which in humans is encoded by the PSAP gene.
Contents
This highly conserved glycoprotein is a precursor for 4 cleavage products: saposins A, B, C, and D. Saposin is an acronym for Sphingolipid Activator PrO[S]teINs. Each domain of the precursor protein is approximately 80 amino acid residues long with nearly identical placement of cysteine residues and glycosylation sites. Saposins A-D localize primarily to the lysosomal compartment where they facilitate the catabolism of glycosphingolipids with short oligosaccharide groups. The precursor protein exists both as a secretory protein and as an integral membrane protein and has neurotrophic activities.
Saposins A-D are required for the hydrolysis of certain sphingolipids by specific lysosomal hydrolases.
Family members
Structure
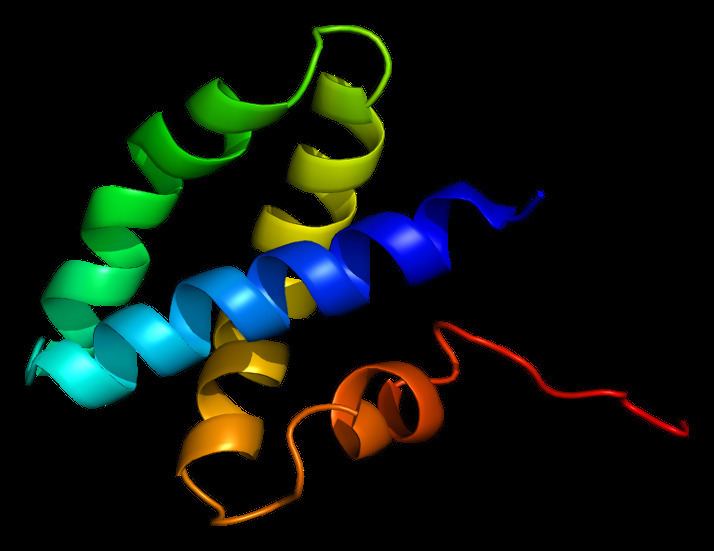
Every saposin contains about 80 amino acid residues and has six equally placed cysteines, two prolines, and a glycosylation site (two in saposin A, one each in saposins B, C, and D). Since saposins characteristics of extreme heat-stability, adundance of disulfide linkages, and resistance to most proteases, they are assumed to be extremely compact and rigidly disulfide-linked molecules. Each saposin has an α-helical structure that is seen as being important for stimulation because this structure is maximal at a pH of 4.5; which is optimal for many lysosomal hydrolases. This helical structure is seen in all (especially with the first region), but saposin has been predicted to have β-sheet configuration due to it first 24 amino acids of the N-end.
Function
They probably act by isolating the lipid substrate from the membrane surroundings, thus making it more accessible to the soluble degradative enzymes. which contains four Saposin-B domains, yielding the active saposins after proteolytic cleavage, and two Saposin-A domains that are removed in the activation reaction. The Saposin-B domains also occur in other proteins, many of them active in the lysis of membranes.
Clinical significance
Mutations in this gene have been associated with Gaucher disease, Tay-Sachs disease, and metachromatic leukodystrophy.
