 | ||
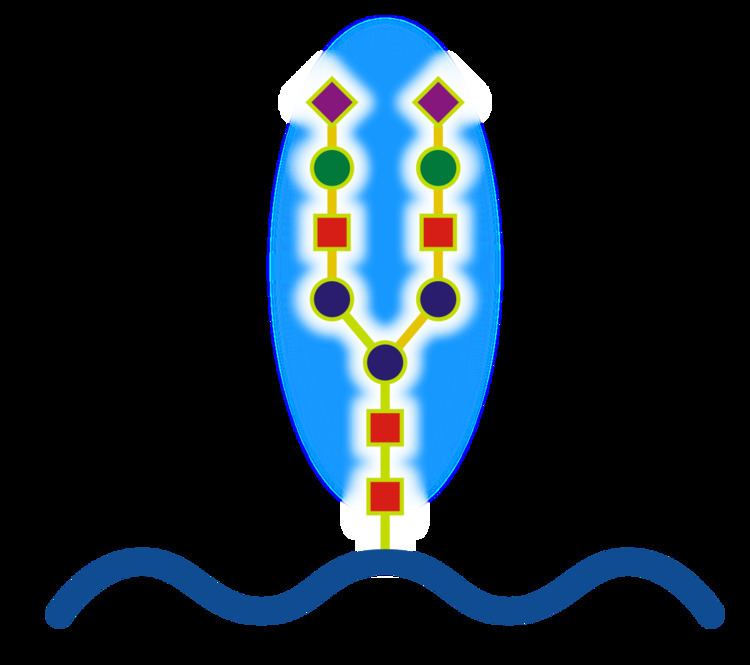
Glycoproteins are proteins that contain oligosaccharide chains (glycans) covalently attached to polypeptide side-chains. The carbohydrate is attached to the protein in a cotranslational or posttranslational modification. This process is known as glycosylation. Secreted extracellular proteins are often glycosylated.
Contents
In proteins that have segments extending extracellularly, the extracellular segments are also often glycosylated. Glycoproteins are also often important integral membrane proteins, where they play a role in cell–cell interactions. It is important to distinguish endoplasmic reticulum-based glycosylation of the secretory system without of reversible cytosolic/nuclear glycosylation. Glycoprotein of the cytosol and nucleus can be modified through the reversible addition of a single GlcNAc residues that is consider reciprocal to phosphorylation and the functions of these are likely to be additional regulatory mechanism that controls phosphorylation-based signalling. In contrast, classical secretory glycosylation can be structurally essential. For example, inhibition of asparagine-linked, i.e. N-linked, glycosylation can prevent glycoprotein folding and full inhibition can be toxic to an individual cell. In contrast, perturbations of terminal processing, which occurs in the Golgi apparatus, is dispensable for isolated cells(as evidence by survival with glycosides inhibitors) but can lead to human disease (Congenital disorders of glycosylation) and can be lethal in animal models. It is therefore likely that the fine processing of glycans is important for endogeneous functionality, such as cell trafficking, but that this is likely to have been secondary to its role in host-pathogen interactions. A famous example of this latter effect is the ABO blood system.
Types of glycosylation
There are several types of glycosylation, although the first two are the most common.
Monosaccharides
Monosaccharides commonly found in eukaryotic glycoproteins include:
The sugar group(s) can assist in protein folding or improve proteins' stability.
Examples
One example of glycoproteins found in the body is mucins, which are secreted in the mucus of the respiratory and digestive tracts. The sugars when attached to mucins give them considerable water-holding capacity and also make them resistant to proteolysis by digestive enzymes.
Glycoproteins are important for white blood cell recognition, especially in mammals. Examples of glycoproteins in the immune system are:
H antigen of the ABO blood compatibility antigens. Other examples of glycoproteins include:
Soluble glycoproteins often show a high viscosity, for example, in egg white and blood plasma.
Variable surface glycoproteins allow the sleeping sickness Trypanosoma parasite to escape the immune response of the host.
The viral spike of the human immunodeficiency virus is heavy glycosylateted. Approximately half the mass of the spike is glycosylation and the glycans act to limit antibody recognition as the glycans are assembled by the host cell and so are largely 'self'. Over time, some patients can evolve antibodies to recognise the HIV glycans and almost all so-called 'broadly neutralising antibodies (bnAbs) recognise some glycans. This is possible mainly because the unusually high density of glycans hinders normal glycan maturation and they are therefore trapped in the premature, high-mannose, state. This provides a window for immune recognition. In addition, as these glycans are much less variable than the underlying protein, they have emerged as promising targets for vaccine design.
Hormones
Hormones that are glycoproteins include:
Analysis
A variety of methods used in detection, purification, and structural analysis of glycoproteins are
