Specialty Infectious disease ICD-9-CM 020 MedlinePlus 000596 | ICD-10 A20.a DiseasesDB 14226 eMedicine med/3381 | |
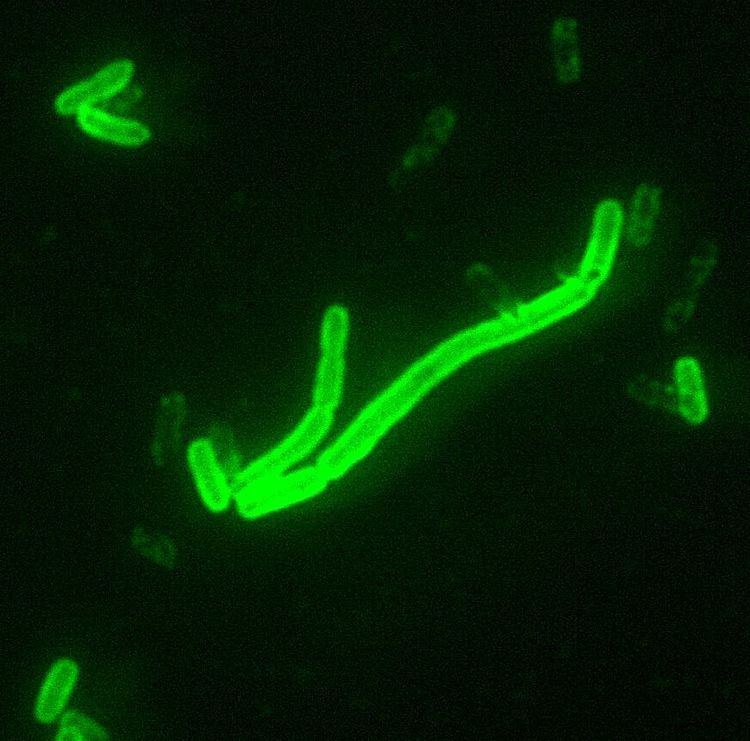 | ||
Plague is an infectious disease that is caused by the bacterium Yersinia pestis. Depending on lung infection, or sanitary conditions, plague can be spread in the air, by direct contact, or very rarely by contaminated undercooked food. The symptoms of plague depend on the concentrated areas of infection in each person: bubonic plague in lymph nodes, septicemic plague in blood vessels, pneumonic plague in lungs. It is treatable if detected early. Plague is still relatively common in some remote parts of the world.
Contents
- Cause
- Bubonic plague
- Septicemic plague
- Pneumonic plague
- Pharyngeal plague
- Meningeal plague
- Other clinical forms
- Treatments
- Plague immunization
- 1994 epidemic in Surat India
- Other contemporary cases
- Antiquity
- Medieval and post medieval pandemics
- First pandemic Early Middle Ages
- Second pandemic from 14th century to 19th century
- Nature of the Black Death
- Third pandemic 19th and 20th centuries
- Plague as a biological weapon
- References
Until June 2007, plague was one of the three epidemic diseases specifically reportable to the World Health Organization (cholera and yellow fever the other two). The bacterium is named after the French-Swiss bacteriologist Alexandre Yersin.
Historically, what are thought to have been massive pandemics of plague swept through Eurasia with very high death rates and causing major cultural changes. The largest of these were the Plague of Justinian of 541–542, The Black Death of the 1340s, continuing in the Second plague pandemic to break out at intervals, and the Third plague pandemic beginning in 1855 and considered inactive from 1959.
The epidemiological use of the term plague is currently applied to any severe bubo inflammation resulting from an infection with Y. pestis. Historically, the medical use of the term plague has been applied to pandemic infections in general. Plague is often synonymous with bubonic plague, but this describes just one of its manifestations. Other names have been used to describe this disease, such as Black Plague and the Black Death; the latter is now used primarily by scholars to describe the second, and most devastating, pandemic of the disease. The etymology of the word plague is believed to come from the Latin word plāga ("blow, wound") and plangere (“to strike, or to strike down”), cf. German Plage (“infestation”).
Cause
Transmission of Y. pestis to an uninfected individual is possible by any of the following means.
Yersinia pestis circulates in animal reservoirs, particularly in rodents, in the natural foci of infection found on all continents except Australia. The natural foci of plague are situated in a broad belt in the tropical and sub–tropical latitudes and the warmer parts of the temperate latitudes around the globe, between the parallels 55 degrees North and 40 degrees South. Contrary to popular belief, rats did not directly start the spread of the bubonic plague. It is mainly a disease in the fleas (Xenopsylla cheopis) that infested the rats, making the rats themselves the first victims of the plague. Infection in a human occurs when a person is bitten by a flea that has been infected by biting a rodent that itself has been infected by the bite of a flea carrying the disease. The bacteria multiply inside the flea, sticking together to form a plug that blocks its stomach and causes it to starve. The flea then bites a host and continues to feed, even though it cannot quell its hunger, and consequently the flea vomits blood tainted with the bacteria back into the bite wound. The bubonic plague bacterium then infects a new victim, and the flea eventually dies from starvation. Serious outbreaks of plague are usually started by other disease outbreaks in rodents, or a rise in the rodent population.
In 1894, two bacteriologists, Alexandre Yersin of France and Kitasato Shibasaburō of Japan, independently isolated the bacterium in Hong Kong responsible for the Third Pandemic. Though both investigators reported their findings, a series of confusing and contradictory statements by Kitasato eventually led to the acceptance of Yersin as the primary discoverer of the organism. Yersin named it Pasteurella pestis in honor of the Pasteur Institute, where he worked, but in 1967 it was moved to a new genus, renamed Yersinia pestis in honor of Yersin. Yersin also noted that rats were affected by plague not only during plague epidemics but also often preceding such epidemics in humans, and that plague was regarded by many locals as a disease of rats: villagers in China and India asserted that, when large numbers of rats were found dead, plague outbreaks soon followed.
In 1898, the French scientist Paul-Louis Simond (who had also come to China to battle the Third Pandemic) established the rat-flea vector that drives the disease. He had noted that persons who became ill did not have to be in close contact with each other to acquire the disease. In Yunnan, China, inhabitants would flee from their homes as soon as they saw dead rats, and on the island of Formosa (Taiwan), residents considered the handling of dead rats heightened the risks of developing plague. These observations led him to suspect that the flea might be an intermediary factor in the transmission of plague, since people acquired plague only if they were in contact with recently dead rats, who had died less than 24 hours before. In a now classic experiment, Simond demonstrated how a healthy rat died of plague, after infected fleas had jumped to it, from a rat which had recently died of the plague.
Bubonic plague
When a flea bites a human and contaminates the wound with regurgitated blood, the plague carrying bacteria are passed into the tissue. Y. pestis can reproduce inside cells, so even if phagocytosed, they can still survive. Once in the body, the bacteria can enter the lymphatic system, which drains interstitial fluid. Plague bacteria secrete several toxins, one of which is known to cause dangerous beta-adrenergic blockade.
Y. pestis spreads through the lymphatic vessels of the infected human until it reaches a lymph node, where it stimulates severe haemorrhagic inflammation that causes the lymph nodes to expand. The expansion of lymph nodes is the cause of the characteristic "bubo" associated with the disease.
If the lymph node is overwhelmed, the infection can pass into the bloodstream, causing secondary septicemic plague and if the lungs are seeded, it can cause secondary pneumonic plague.
Septicemic plague
Lymphatics ultimately drain into the bloodstream, so the plague bacteria may enter the blood and travel to almost any part of the body. In septicemic plague, bacterial endotoxins cause disseminated intravascular coagulation (DIC), causing tiny clots throughout the body and possibly ischaemic necrosis (tissue death due to lack of circulation/perfusion to that tissue) from the clots. DIC results in depletion of the body's clotting resources, so that it can no longer control bleeding. Consequently, there is bleeding into the skin and other organs, which can cause red and/or black patchy rash and hemoptysis/hematemesis (coughing up/ vomiting of blood). There are bumps on the skin that look somewhat like insect bites; these are usually red, and sometimes white in the center. Untreated, septicemic plague is usually fatal. Early treatment with antibiotics reduces the mortality rate to between 4 and 15 percent. People who die from this form of plague often die on the same day symptoms first appear.
Pneumonic plague
The pneumonic form of plague arises from infection of the lungs. It causes coughing and sneezing and thereby produces airborne droplets that contain bacterial cells and are likely to infect anyone inhaling them. The incubation period for pneumonic plague is short, usually two to four days, but sometimes just a few hours. The initial signs are indistinguishable from several other respiratory illnesses; they include headache, weakness, and hemoptysis or hematemesis (spitting or vomiting of blood). The course of the disease is rapid; unless diagnosed and treated soon enough, typically within a few hours, death may follow in one to six days; in untreated cases mortality is nearly 100%.
Pharyngeal plague
This is an uncommon form of plague that resembles tonsillitis found in cases of close contact of patients with other forms of plague.
Meningeal plague
This form of plague occurs when bacteria cross the blood-brain barrier, leading to infectious meningitis.
Other clinical forms
There are a few other rare manifestations of plague, including asymptomatic plague and abortive plague. Cellulocutaneous plague sometimes results in infection of the skin and soft tissue, often around the bite site of a flea.
Treatments
Waldemar Haffkine, a doctor who worked in Bombay, India, was the first to invent and test a plague vaccine against bubonic plague in 1897.
If diagnosed in time the various forms of plague are usually highly responsive to antibiotic therapy. The antibiotics often used are streptomycin, chloramphenicol and tetracycline. Amongst the newer generation of antibiotics, gentamicin and doxycycline have proven effective in monotherapeutic treatment of plague.
The plague bacterium could develop drug-resistance and again become a major health threat. One case of a drug-resistant form of the bacterium was found in Madagascar in 1995. A further outbreak in Madagascar was reported in November 2014.
Plague immunization
Since human plague is rare in most parts of the world, routine vaccination is not needed other than for those at particularly high risk of exposure, nor for people living in areas with enzootic plague, meaning it occurs at regular, predictable rates in populations and specific areas, such as the western United States. It is not even indicated for most travellers to countries with known recent reported cases, particularly if their travel is limited to urban areas with modern hotels. The CDC thus only recommends vaccination for: (1) all laboratory and field personnel who are working with Y. pestis organisms resistant to antimicrobials; (2) people engaged in aerosol experiments with Y. pestis; and (3) people engaged in field operations in areas with enzootic plague where preventing exposure is not possible (such as some disaster areas).
A systematic review by the Cochrane Collaboration found no studies of sufficient quality to make any statement on the efficacy of the vaccine.
1994 epidemic in Surat, India
In 1994, there was a pneumonic plague epidemic in Surat, India that resulted in 52 deaths and in a large internal migration of about 300,000 residents, who fled fearing quarantine.
A combination of heavy monsoon rain and clogged sewers led to massive flooding which resulted in unhygienic conditions and a number of uncleared animal carcasses. It is believed that this situation precipitated the epidemic. There was widespread fear that the sudden rush of people from this area might spread the epidemic to other parts of India and the world, but that scenario was averted, probably as a result of effective public health response mounted by the Indian health authorities. Some countries, especially those in the nearby Gulf region, took the step of cancelling some flights and putting a pause on shipments from India.
Much like the Black Death that spread through medieval Europe, some questions still remain unanswered about the 1994 epidemic in Surat.
Initial questions about whether it was an epidemic of plague arose because the Indian health authorities were unable to culture Yersinia pestis, but this could have been due to poor laboratory procedures. Yet several lines of evidence strongly suggest that it was a plague epidemic: blood tests for Yersinia were positive, a number of individuals showed antibodies against Yersinia and the clinical symptoms displayed by the affected were all consistent with the disease being plague.
Other contemporary cases
Antiquity
Plasmids of Y. pestis have been detected in archaeological samples of the teeth of seven Bronze Age individuals from 5000 years ago (3000 BC), in the Afanasievo culture in Siberia, the Corded Ware culture in Estonia, the Sintashta culture in Russia, the Unetice culture in Poland and the Andronovo culture in Siberia. Y. Pestis existed over Eurasia during the Bronze Age. Estimates of the age of the Most recent common ancestor of all Y. Pestis is estimated at 5,783 years Before Present.
The Yersinia murine toxin (ymt) allows the bacteria to infect fleas, which can then transmit bubonic plague. Early ancestral versions of Y. Pestis did not have the ymt gene, which was only detected in a 951 calibrated BC sample.
The Amarna letters and the Plague Prayers of Mursili II describe an outbreak of a disease among the Hittites, though some modern sources say it may be Tularemia. The First Book of Samuel describes a possible plague outbreak in Philistia, and the Septuagint version says it was caused by a "ravaging of mice".
In the second year of the Peloponnesian War (430 BC), Thucydides described an epidemic disease which was said to have begun in Ethiopia, passed through Egypt and Libya, then come to the Greek world. In the Plague of Athens, the city lost possibly one third of its population, including Pericles. Modern historians disagree on whether the plague was a critical factor in the loss of the war. Although this epidemic has long been considered an outbreak of plague, many modern scholars believe that typhus, smallpox, or measles may better fit the surviving descriptions. A recent study of DNA found in the dental pulp of plague victims suggests that typhoid was actually responsible.
In the first century AD, Rufus of Ephesus, a Greek anatomist, refers to an outbreak of plague in Libya, Egypt, and Syria. He records that Alexandrian doctors named Dioscorides and Posidonius described symptoms including acute fever, pain, agitation, and delirium. Buboes—large, hard, and non-suppurating—developed behind the knees, around the elbows, and "in the usual places." The death toll of those infected was very high. Rufus also wrote that similar buboes were reported by a Dionysius Curtus, who may have practiced medicine in Alexandria in the third century BC. If this is correct, the eastern Mediterranean world may have been familiar with bubonic plague at that early date.
In the second century, the Antonine Plague, named after Marcus Aurelius’ family name of Antoninus and also known as the Plague of Galen, who had first hand knowledge of the disease, may in fact have been smallpox. Galen was in Rome when it struck in 166 AD, and was also present in the winter of 168–69 during an outbreak among troops stationed at Aquileia; he had experience with the epidemic, referring to it as very long lasting, and describes its symptoms and his treatment of it. Unfortunately, his references are scattered and brief. According to Barthold Georg Niebuhr "this pestilence must have raged with incredible fury; it carried off innumerable victims. The ancient world never recovered from the blow inflected upon it by the plague which visited it in the reign of M. Aurelius." The mortality rate of the plague was 7–10 percent; the outbreak in 165–6–168 would have caused approximately 3.5 to 5 million deaths. Otto Seek believes that over half the population of the empire perished. J. F. Gilliam believes that the Antonine plague probably caused more deaths than any other epidemic during the empire before the mid-3rd century.
Medieval and post-medieval pandemics
Local outbreaks of the plague are grouped into three plague pandemics, whereby the respective start and end dates and the assignment of some outbreaks to either pandemic are still subject to discussion. According to Joseph P. Byrne from Belmont University, the pandemics were:
However, the late medieval Black Death is sometimes seen not as the start of the second, but as the end of the first pandemic – in that case, the second pandemic's start would be 1361; also vary the end dates of the second pandemic given in literature, e.g. ~1890 instead of ~1840.
First pandemic: Early Middle Ages
The Plague of Justinian in AD 541–542 is the first known attack on record, and marks the first firmly recorded pattern of bubonic plague. This disease is thought to have originated in China. It then spread to Africa from where the huge city of Constantinople imported massive amounts of grain, mostly from Egypt, to feed its citizens. The grain ships were the source of contagion for the city, with massive public granaries nurturing the rat and flea population. At its peak, Procopius said the plague was killing 10,000 people in Constantinople every day. The real number was more likely close to 5,000 a day. The plague ultimately killed perhaps 40% of the city's inhabitants, and then continued to kill up to a quarter of the human population of the eastern Mediterranean.
In AD 588 a second major wave of plague spread through the Mediterranean into what is now France. It is estimated that the Plague of Justinian killed as many as 100 million people across the world. It caused Europe's population to drop by around 50% between 541 and 700. It also may have contributed to the success of the Arab conquests. An outbreak of it in the AD 560s was described in AD 790 as causing "swellings in the glands ... in the manner of a nut or date" in the groin "and in other rather delicate places followed by an unbearable fever". While the swellings in this description have been identified by some as buboes, there is some contention as to whether the pandemic should be attributed to the bubonic plague, Yersinia pestis, known in modern times.
Second pandemic: from 14th century to 19th century
From 1347 to 1351, the Black Death, a massive and deadly pandemic originating in China, spread along the Silk Road and swept through Asia, Europe and Africa. It may have reduced the world's population from 450 million to between 350 and 375 million. China lost around half of its population, from around 123 million to around 65 million; Europe around 1⁄3 of its population, from about 75 million to about 50 million; and Africa approximately 1⁄8 of its population, from around 80 million to 70 million (mortality rates tended to be correlated with population density so Africa, being less dense overall, had the lowest death rate). This makes the Black Death the largest death toll from any known non-viral epidemic. Although accurate statistical data does not exist, it is thought that 1.4 million died in England ( 1⁄3 of England's 4.2 million people), while an even higher percentage of Italy's population was likely wiped out. On the other hand, north-eastern Germany, Bohemia, Poland and Hungary are believed to have suffered less, and there are no estimates available for Russia or the Balkans. It is conceivable that Russia may not have been as affected due to its very cold climate and large size, hence often less close contact with the contagion.
The plague repeatedly returned to haunt Europe and the Mediterranean throughout the 14th to 17th centuries. According to Biraben, plague was present somewhere in Europe in every year between 1346 and 1671. The Second Pandemic was particularly widespread in the following years: 1360–1363; 1374; 1400; 1438–1439; 1456–1457; 1464–1466; 1481–1485; 1500–1503; 1518–1531; 1544–1548; 1563–1566; 1573–1588; 1596–1599; 1602–1611; 1623–1640; 1644–1654; and 1664–1667; subsequent outbreaks, though severe, marked the retreat from most of Europe (18th century) and northern Africa (19th century). According to Geoffrey Parker, "France alone lost almost a million people to plague in the epidemic of 1628–31."
In England, in the absence of census figures, historians propose a range of pre-incident population figures from as high as 7 million to as low as 4 million in 1300, and a postincident population figure as low as 2 million. By the end of 1350, the Black Death subsided, but it never really died out in England. Over the next few hundred years, further outbreaks occurred in 1361–62, 1369, 1379–83, 1389–93, and throughout the first half of the 15th century. An outbreak in 1471 took as much as 10–15% of the population, while the death rate of the plague of 1479–80 could have been as high as 20%. The most general outbreaks in Tudor and Stuart England seem to have begun in 1498, 1535, 1543, 1563, 1589, 1603, 1625, and 1636, and ended with the Great Plague of London in 1665.
In 1466, perhaps 40,000 people died of plague in Paris. During the 16th and 17th centuries, plague visited Paris for almost one year out of three. The Black Death ravaged Europe for three years before it continued on into Russia, where the disease hit somewhere once every five or six years from 1350 to 1490. Plague epidemics ravaged London in 1563, 1593, 1603, 1625, 1636, and 1665, reducing its population by 10 to 30% during those years. Over 10% of Amsterdam's population died in 1623–1625, and again in 1635–1636, 1655, and 1664. There were 22 outbreaks of plague in Venice between 1361 and 1528. The plague of 1576–1577 killed 50,000 in Venice, almost a third of the population. Late outbreaks in central Europe included the Italian Plague of 1629–1631, which is associated with troop movements during the Thirty Years' War, and the Great Plague of Vienna in 1679. Over 60% of Norway's population died from 1348 to 1350. The last plague outbreak ravaged Oslo in 1654.
In the first half of the 17th century, the Great Plague of Milan claimed some 1.7 million victims in Italy, or about 14% of the population. In 1656, the plague killed about half of Naples' 300,000 inhabitants. More than 1.25 million deaths resulted from the extreme incidence of plague in 17th-century Spain. The plague of 1649 probably reduced the population of Seville by half. In 1709–1713, a plague epidemic that followed the Great Northern War (1700–1721, Sweden v. Russia and allies) killed about 100,000 in Sweden, and 300,000 in Prussia. The plague killed two-thirds of the inhabitants of Helsinki, and claimed a third of Stockholm's population. Western Europe's last major epidemic occurred in 1720 in Marseilles, in Central Europe the last major outbreaks happened during the plague during the Great Northern War, and in Eastern Europe during the Russian plague of 1770–72.
The Black Death ravaged much of the Islamic world. Plague was present in at least one location in the Islamic world virtually every year between 1500 and 1850. Plague repeatedly struck the cities of North Africa. Algiers lost 30,000–50,000 to it in 1620–21, and again in 1654–57, 1665, 1691, and 1740–42. Plague remained a major event in Ottoman society until the second quarter of the 19th century. Between 1701 and 1750, 37 larger and smaller epidemics were recorded in Constantinople, and 31 between 1751 and 1800. Baghdad has suffered severely from visitations of the plague, and sometimes two-thirds of its population has been wiped out.
Nature of the Black Death
In the early 20th century, following the identification by Yersin and Kitasato of the plague bacterium that caused the late 19th and early 20th century Asian bubonic plague (the Third Pandemic), most scientists and historians came to believe that the Black Death was an incidence of this plague, with a strong presence of the more contagious pneumonic and septicemic varieties increasing the pace of infection, spreading the disease deep into inland areas of the continents.
Some modern researchers have argued that the disease was more likely to have been viral, pointing to the absence of rats from some parts of Europe that were badly affected and to the conviction of people at the time that the disease was spread by direct human contact. According to the accounts of the time the Black Death was extremely virulent, unlike the 19th and early 20th century bubonic plague. Samuel K. Cohn has made a comprehensive attempt to rebut the bubonic plague theory. Researchers have offered a mathematical model based on the changing demography of Europe from AD 1000 to 1800 demonstrating how plague epidemics, 1347 to 1670, could have provided the selection pressure that raised the frequency of a mutation to the level seen today that prevent HIV from entering macrophages and CD4+ T cells that carry the mutation (the average frequency of this allele is 10% in European populations). It is suggested that the original single mutation appeared over 2,500 years ago and that persistent epidemics of a hemorrhagic fever struck at the early classical civilizations.
However, there is evidence that two previously unknown clades (variant strains) of Y. pestis were responsible for the Black Death. A multinational team conducted new surveys that used both ancient DNA analyses and protein-specific detection to find DNA and protein signatures specific for Y. pestis in human skeletons from widely distributed mass graves in northern, central and southern Europe that were associated archaeologically with the Black Death and subsequent resurgences. The authors concluded that this research, together with prior analyses from the south of France and Germany,
"... ends the debate about the etiology of the Black Death, and unambiguously demonstrates that Y. pestis was the causative agent of the epidemic plague that devastated Europe during the Middle Ages."The study also identified two previously unknown but related strains of Y. pestis that were associated with distinct medieval mass graves. These were found to be ancestral to modern isolates of the present-day Y. pestis strains 'Orientalis' and 'Medievalis', suggesting that these variant strains (which are now presumed to be extinct) may have entered Europe in two waves. Surveys of plague pit remains in France and England indicate that the first variant entered Europe through the port of Marseille around November 1347 and spread through France over the next two years, eventually reaching England in the spring of 1349, where it spread through the country in three successive epidemics.
Surveys of plague pit remains from the Netherlands town of Bergen op Zoom showed evidence of a second Y. pestis genotype which differed from that found in Britain and France and this second strain is now thought to have been responsible for the pandemic that spread through the Low Countries from 1350. This discovery implies that Bergen op Zoom (and possibly other parts of the southern Netherlands) was not directly infected from England or France c. AD 1349, and the researchers have suggested that a second wave of plague infection, distinct from that which occurred in Britain and France, may have been carried to the Low Countries from Norway, the Hanseatic cities, or another site.
Third pandemic: 19th and 20th centuries
The Third Pandemic began in China's Yunnan province in 1855, spreading plague to all inhabited continents and ultimately killing more than 12 million people in India and China alone. Casualty patterns indicate that waves of this pandemic may have come from two different sources. The first was primarily bubonic and was carried around the world through ocean-going trade, transporting infected persons, rats, and cargoes harboring fleas. The second, more virulent strain was primarily pneumonic in character, with a strong person-to-person contagion. This strain was largely confined to Manchuria and Mongolia. Researchers during the "Third Pandemic" identified plague vectors and the plague bacterium (see above), leading in time to modern treatment methods.
Plague occurred in Russia in 1877–1889 in rural areas near the Ural Mountains and the Caspian Sea. Efforts in hygiene and patient isolation reduced the spread of the disease, with approximately 420 deaths in the region. Significantly, the region of Vetlianka in this area is near a population of the bobak marmot, a small rodent considered a very dangerous plague reservoir. The last significant Russian outbreak of Plague was in Siberia in 1910 after sudden demand for marmot skins (a substitute for sable) increased the price by 400 percent. The traditional hunters would not hunt a sick Marmot and it was taboo to eat the fat from under the arm (the axillary lymphatic gland that often harboured the plague) so outbreaks tended to be confined to single individuals. The price increase, however, attracted thousands of Chinese hunters from Manchuria who not only caught the sick animals but also ate the fat, which was considered a delicacy. The plague spread from the hunting grounds to the terminus of the Chinese Eastern Railway and then followed the track for 2,700 km. The plague lasted 7 months and killed 60,000 people.
The bubonic plague continued to circulate through different ports globally for the next fifty years; however, it was primarily found in Southeast Asia. An epidemic in Hong Kong in 1894 had particularly high death rates, 90%. As late as 1897, medical authorities in the European powers organized a conference in Venice, seeking ways to keep the plague out of Europe. Mumbai plague epidemic struck the city of Bombay (Mumbai) in 1896. The disease reached the Territory of Hawaii in December 1899, and the Board of Health's decision to initiate controlled burns of select buildings in Honolulu's Chinatown turned into an uncontrolled fire which led to the inadvertent burning of most of Chinatown on January 20, 1900. Shortly thereafter, plague reached the continental US, initiating the San Francisco plague of 1900–1904. Plague persisted in Hawaii on the outer islands of Maui and Hawaii (The Big Island) until it was finally eradicated in 1959.
Although the outbreak that began in China in 1855 is conventionally known as the Third Pandemic (see above), it is unclear whether there have been fewer, or more, than three major outbreaks of bubonic plague. Most modern outbreaks of bubonic plague amongst humans have been preceded by a striking, high mortality amongst rats, yet this phenomenon is absent from descriptions of some earlier plagues, especially the Black Death. The buboes, or swellings in the groin, that are especially characteristic of bubonic plague, are a feature of other diseases as well.
Research done by a team of biologists from the Institute of Pasteur in Paris and Johannes Gutenberg University Mainz in Germany by analyzing the DNA and proteins from plague pits was published in October 2010, reported beyond doubt that all 'the three major plagues' were due to at least two previously unknown strains of Yersinia pestis and originated from China. A team of medical geneticists led by Mark Achtman of University College Cork in Ireland reconstructed a family tree of the bacterium and concluded in an online issue of Nature Genetics published on 31 October 2010 that all three of the great waves of plague originated from China.
Plague as a biological weapon
Plague has a long history as a biological weapon. Historical accounts from ancient China and medieval Europe detail the use of infected animal carcasses, such as cows or horses, and human carcasses, by the Xiongnu/Huns, Mongols, Turks, and other groups, to contaminate enemy water supplies. Han Dynasty General Huo Qubing is recorded to have died of such a contamination while engaging in warfare against the Xiongnu. Plague victims were also reported to have been tossed by catapult into cities under siege.
In 1347, the Genoese possession of Caffa, a great trade emporium on the Crimean peninsula, came under siege by an army of Mongol warriors of the Golden Horde under the command of Janibeg. After a protracted siege during which the Mongol army was reportedly withering from the disease, they decided to use the infected corpses as a biological weapon. The corpses were catapulted over the city walls, infecting the inhabitants. The Genoese traders fled, transferring the plague (Black Death) via their ships into the south of Europe, hence its rapid spread.
During World War II, the Japanese Army developed weaponised plague, based on the breeding and release of large numbers of fleas. During the Japanese occupation of Manchuria, Unit 731 deliberately infected Chinese, Korean, and Manchurian civilians and prisoners of war with the plague bacterium. These subjects, termed "maruta", or "logs", were then studied by dissection, others by vivisection while still conscious. Members of the unit such as Shiro Ishii were exonerated from the Tokyo tribunal by Douglas MacArthur but 12 of them were prosecuted in the Khabarovsk War Crime Trials in 1949 during which some admitted having spread bubonic plague within a 36-km radius around the city of Changde.
Ishii innovated bombs containing live mice and fleas, with very small explosive loads, to deliver the weaponized microbes, overcoming the problem of the explosive killing the infected animal and insect by the use of a ceramic, rather than metal, casing for the warhead. While no records survive of the actual usage of the ceramic shells, prototypes exist and are believed to have been used in experiments during WWII.
After World War II, both the United States and the Soviet Union developed means of weaponising pneumonic plague. Experiments included various delivery methods, vacuum drying, sizing the bacterium, developing strains resistant to antibiotics, combining the bacterium with other diseases (such as diphtheria), and genetic engineering. Scientists who worked in USSR bio-weapons programs have stated that the Soviet effort was formidable and that large stocks of weaponised plague bacteria were produced. Information on many of the Soviet projects is largely unavailable. Aerosolized pneumonic plague remains the most significant threat.
The plague can be easily treated with antibiotics, which some countries, such as the United States, have large supplies on hand if such an attack should occur, thus making the threat less severe.
