 | ||
Opioid receptors part 1
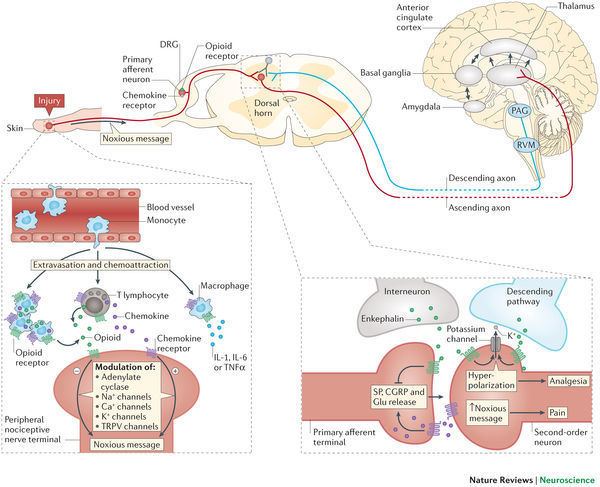
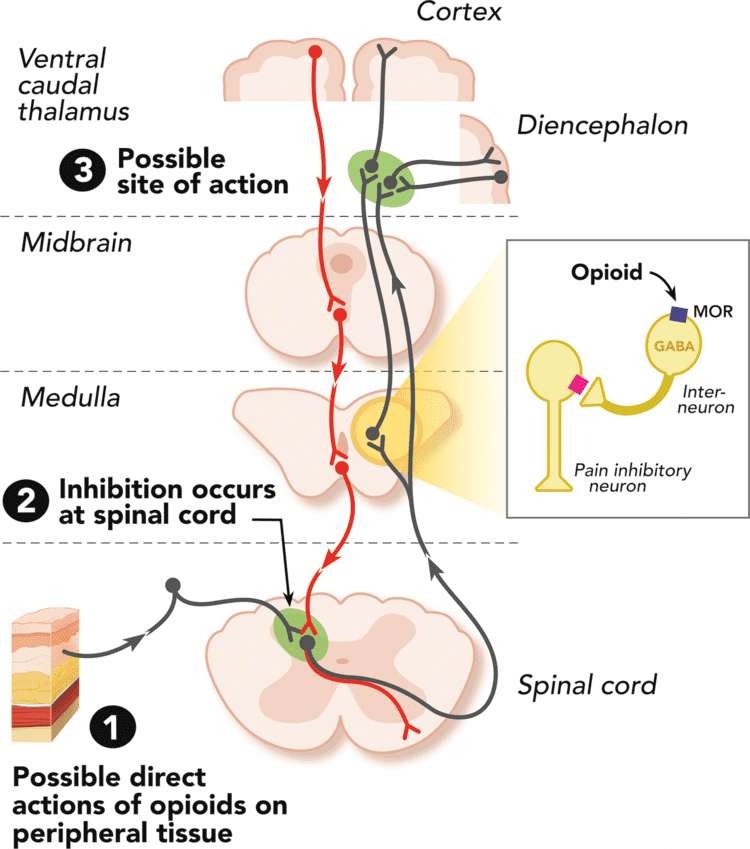
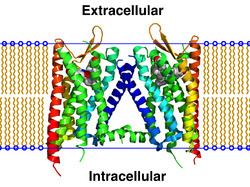
Opioid receptors are a group of inhibitory G protein-coupled receptors with opioids as ligands. The endogenous opioids are dynorphins, enkephalins, endorphins, endomorphins and nociceptin. The opioid receptors are ~40% identical to somatostatin receptors (SSTRs). Opioid receptors are distributed widely in the brain, and are found in the spinal cord and digestive tract.
Contents
- Opioid receptors part 1
- Discovery
- Purification
- Major subtypes
- Evolution
- Naming
- Additional receptors
- opioid receptor
- Pathology
- Receptor heteromers
- References
Discovery
By the mid-1960s, it had become apparent from pharmacologic studies that opiate drugs were likely to exert their actions at specific receptor sites, and that there were likely to be multiple such sites. Early studies had indicated that opiates appeared to accumulate in the brain. The receptors were first identified as specific molecules through the use of binding studies, in which opiates that had been labeled with radioisotopes were found to bind to brain membrane homogenates. The first such study was published in 1971, using 3H-levorphanol. In 1973, Candace Pert and Solomon H. Snyder published the first detailed binding study of what would turn out to be the μ opioid receptor, using 3H-naloxone. That study has been widely credited as the first definitive finding of an opioid receptor, although two other studies followed shortly after.
Purification
Purification of the receptor further verified its existence. The first attempt to purify the receptor involved the use of a novel opioid receptor antagonist called chlornaltrexamine that was demonstrated to bind to the opioid receptor. Caruso later purified the detergent-extracted component of rat brain membrane that eluted with the specifically bound 3H-chlornaltrexamine.
Major subtypes
There are four major subtypes of opioid receptors:
(I). Name based on order of discovery
Evolution
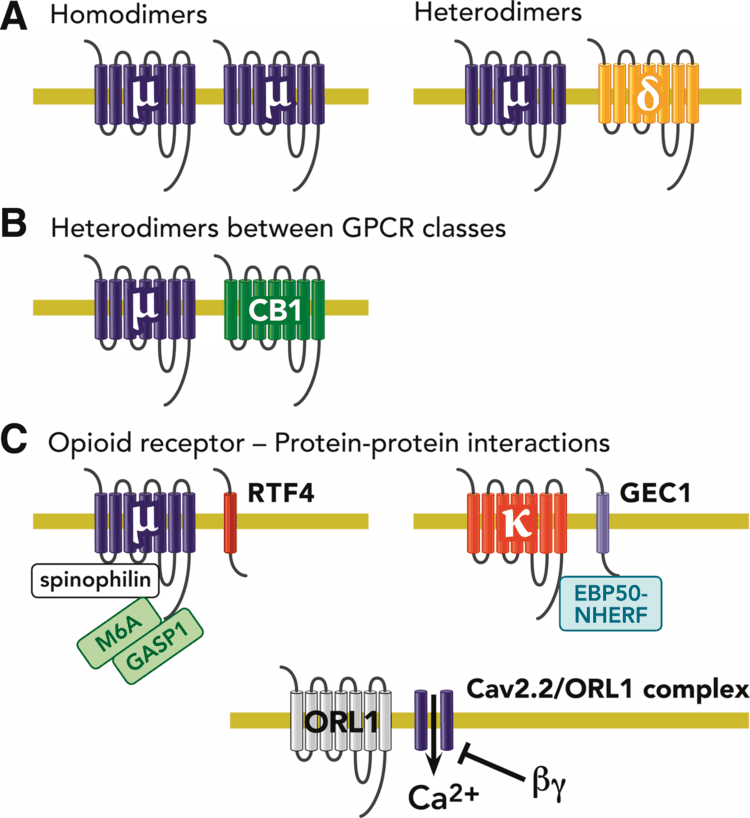
The opioid receptor family originated from two duplication events of a single ancestral opioid receptor early in vertebrate evolution. Phylogenetic analysis demonstrates that the family of opioid receptors was already present at the origin of jawed vertebrates over 450 million years ago. In humans, this paralogon resulting from a double tetraploidization event resulted in the receptor genes being located on chromosomes 1, 6, 8, and 20. Tetraploidization events often result in the loss of one or more of the duplicated genes, but in this case, nearly all species retain all four opioid receptors, indicating important and specific function. The receptor families delta, kappa, and mu demonstrate 55–58% identity to one another, and a 48–49% homology to the nociceptin receptor. Taken together, this indicates that the NOP receptor gene, OPRL1, has equal evolutionary origin, but a higher mutation rate, than the other receptor genes.
The endogenous opioid system is thought to be important in mediating complex social behaviors involved in the formation of stable, emotionally committed relationships. Social attachment was demonstrated to be mediated by the opioid system through experiments administering morphine and naltrexone, an opioid agonist and antagonist, to juvenile guinea pigs. The agonist decreased the preference of the juvenile to be near the mother and reduced distress vocalization whereas the antagonist had the opposite effects. Experiments were corroborated in dogs, chicks, and rats confirming the evolutionary importance of opioid signaling in these behaviors. Researchers have also found that systemic naltrexone treatment of female prairie voles during initial exposure to a male reduced subsequent mating bouts and nonsexual socialization with this familiar partner, when a choice test including a novel male was performed afterwards. This points to a role for opioid receptors in mating behaviors.
There is evidence that human specific cognitive traits rely not on coding differences for the receptors or ligands, which display 99% homology with primates, but instead due to regulatory changes in expression levels that are specifically selected for.
Naming
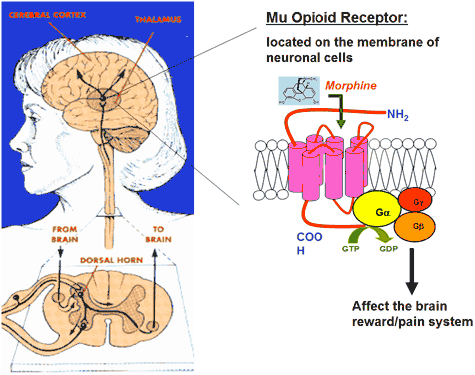
The receptors were named using the first letter of the first ligand that was found to bind to them. Morphine was the first chemical shown to bind to mu receptors. The first letter of the drug morphine is m, rendered as the corresponding Greek letter μ. In similar manner, a drug known as ketocyclazocine was first shown to attach itself to κ receptors, while the δ receptor was named after the mouse vas deferens tissue in which the receptor was first characterised. An additional opioid receptor was later identified and cloned based on homology with the cDNA. This receptor is known as the nociceptin receptor or ORL1 (opiate receptor-like 1).
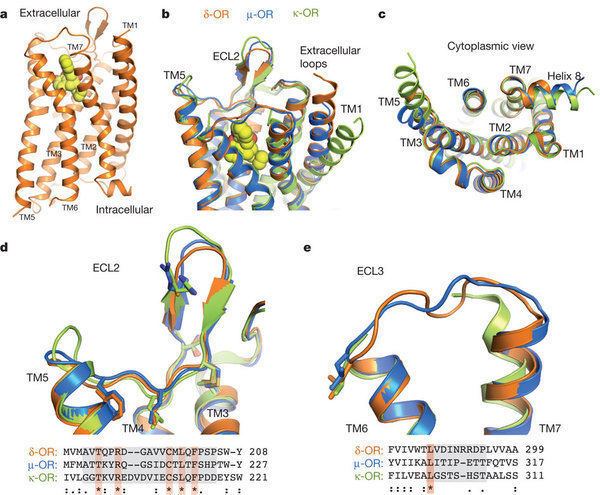
The opioid receptor types are ~70% identical with differences located at N and C termini. The μ receptor is perhaps the most important. It is thought that the G protein binds to the third intracellular loop of the opioid receptors. Both in mice and humans, the genes for the various receptor subtypes are located on different chromosomes.
Separate subtypes have been identified in human tissue. Research has so far failed to identify the genetic evidence of the subtypes, and it is thought that they arise from post-translational modification of cloned receptor types.
An IUPHAR subcommittee has recommended that appropriate terminology for the 3 classical (μ, δ, κ) receptors, and the non-classical (nociceptin) receptor, should be MOP, DOP, KOP and NOP respectively.
Additional receptors
Sigma (σ) receptors were once considered to be opioid receptors due to the antitussive actions of many opioid drugs' being mediated via σ receptors, and the first selective σ agonists being derivatives of opioid drugs (e.g., allylnormetazocine). However, σ receptors were found to not be activated by endogenous opioid peptides, and are quite different from the other opioid receptors in both function and gene sequence, so they are now not usually classified with the opioid receptors.
The existence of further opioid receptors (or receptor subtypes) has also been suggested because of pharmacological evidence of actions produced by endogenous opioid peptides, but shown not to be mediated through any of the four known opioid receptor subtypes. The existence of receptor subtypes or additional receptors other than the classical opioid receptors (μ, δ, κ) has been based on limited evidence, since only three genes for the three main receptors have been identified. The only one of these additional receptors to have been definitively identified is the zeta (ζ) opioid receptor, which has been shown to be a cellular growth factor modulator with met-enkephalin being the endogenous ligand. This receptor is now most commonly referred to as the opioid growth factor receptor (OGFr).
ε opioid receptor
Another postulated opioid receptor is the ε opioid receptor. The existence of this receptor was suspected after the endogenous opioid peptide beta-endorphin was shown to produce additional actions that did not seem to be mediated through any of the known opioid receptors. Activation of this receptor produces strong analgesia and release of met-enkephalin, and a number of widely used opioid agonists such as the μ agonist etorphine and the κ agonist bremazocine have been shown to act as agonists for this effect (even in the presence of antagonists to their more well known targets), while buprenorphine has been shown to act as an epsilon antagonist. Several selective agonists and antagonists are now available for the putative epsilon receptor, however efforts to locate a gene for this receptor have been unsuccessful, and epsilon-mediated effects were absent in μ/δ/κ "triple knockout" mice, suggesting the epsilon receptor is likely to be either a splice variant derived from alternate post-translational modification, or a heteromer derived from hybridization of two or more of the known opioid receptors.
Pathology
Some forms of mutations in δ-opioid receptors have resulted in constant receptor activation.
