Scientific name Nitrobacter | Higher classification Nitrobacteraceae | |
 | ||
Similar Nitrosomonas, Nitrosococcus, Nitrospira, Azotobacter, Thiobacillus | ||
Nitrosomonas and nitrobacter
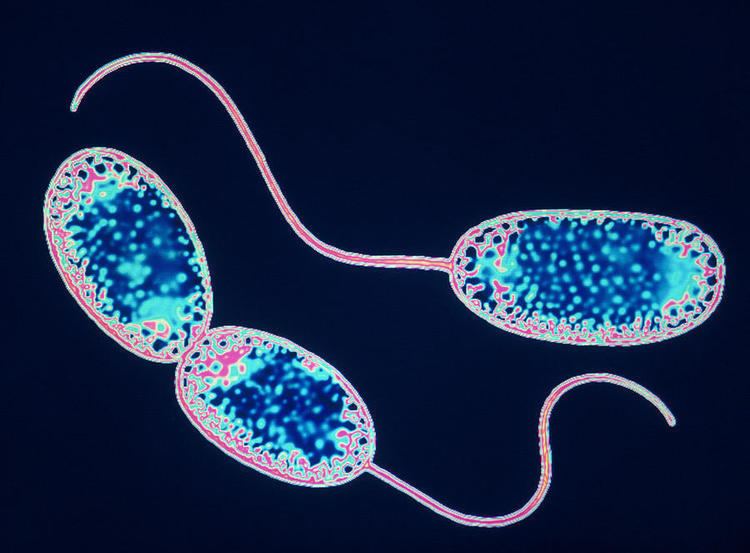
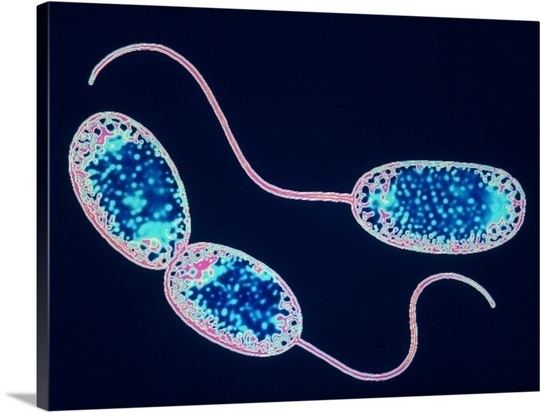
Nitrobacter is a genus of mostly rod-shaped, gram-negative, and chemoautotrophic Bacteria. They are non-motile and reproduce via budding. Cells may also undergo binary fission. Nitrobacter cells may or may not be motile. They are obligate aerobes and have a doubling time of about 13 hours.
Contents
- Nitrosomonas and nitrobacter
- The Nitro Bros Nitrosomonas and Nitrobacter
- Etymology
- Morphology
- Metabolism and Growth
- Ecology and Distribution
- History
- Main Species
- References
Nitrobacter play an important role in the nitrogen cycle by oxidizing nitrite into nitrate in soil and marine systems. Unlike plants, where electron transfer in photosynthesis provides the energy for carbon fixation, Nitrobacter uses energy from the oxidation of Nitrite ions, NO2−, into Nitrate ions, NO3−, to fulfill their energy needs. Nitrobacter fixes carbon dioxide via the Calvin cycle for their carbon requirements. Nitrobacter cells have been show to recover following extreme CO2 exposure
Nitrobacter have an optimum pH between 7.3 and 7.5, and will die in temperatures exceeding 120 °F (49 °C) or below 32 °F (0 °C).
According to Grundmann, Nitrobacter seem to grow optimally at 38 °C and at a pH of 7.9, but Holt states that Nitrobacter grow optimally at 28 °C and grows within a pH range of 5.8 -8.5 and has an pH optima between 7.6 and 7.8. Nitrobacter belongs to the α-subclass of the Proteobacteria.
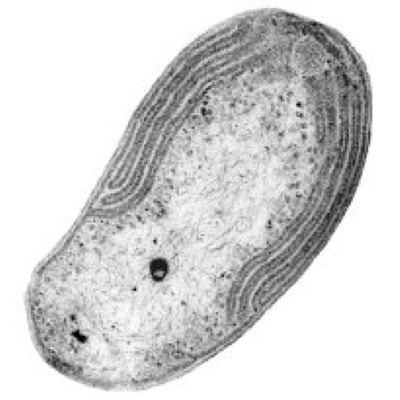
The Nitro Bros - Nitrosomonas and Nitrobacter
Etymology
The name Nitrobacter derives from the Latin neuter gender noun nitrum, nitri, alkalis; the Ancient Greek noun βακτηρία, βακτηρίᾱς, rod.
Morphology
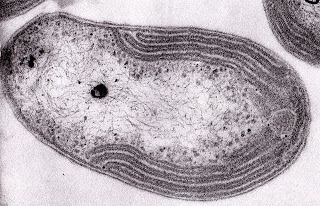
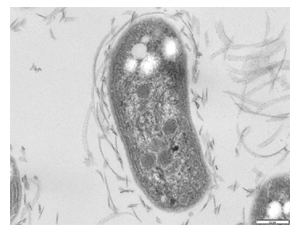
Nitrobacter are gram-negative bacteria and may either be rod shaped, pear-shaped or pleomorphic. They are typically 0.5-0.9 x 1.0-2.0μm in size and have a intracytomembrane polar cap. Due to the presence of cytochromes, they are often yellow in cell suspensions. The nitrate oxydising system on membranes is cytoplasmic.
Metabolism and Growth
Nitrobacter may reproduce by budding or binary fission. Carboxysomes which aid carbon fixation are found in lithoautotrophically and mixotrophically grown cells. Additional energy conserving inclusions are PHB granules and polyphosphates. When both nitrite and organic substances are present, cells can exhibit biphasic growth, first the nitrite is used and after a lag phase, organic matter is oxidized. Chemoorganotrophic growth is slow and unbalanced thus more poly-β- hydroxybutyrate granules are seen that distort the shape and size of the cells.
Ecology and Distribution
Nitrobacter play an essential role in aquaponics. Nitrobacter has a wide distribution as it occupies both aquatic and land environments across the different species in the genus. nitrifying bacteria have an optimum growth between 25-30 °C, and cannot survive past the upper limit of 49 °C or the lower limit of 0 °C, therefore limiting its distribution even though it encompasses a variety of habitats. Its primary ecological role is to oxidize nitrite to nitrate which is readily absorbed by plants, after Nitrosomonas bacteria first convert ammonia into nitrites and since all Nitrobacter are obligate aerobes, oxygen along with phosphorous tend to be the limiting factors preventing them from being able to efficiently fix nitrogen. The greater impact of nitrification and Nitrobacter in both oceanic and terrestrial ecosystems would be its effect on the process of eutrophication. The distribution and differences in N-fixing rate across the Nitrobacter genus may be attributed to differences in the plasmids amongst the species , as data presented in Schutt (1990) imply habitat specific plasmid DNA induced by adaptation for some of the lakes that were investigated. A follow up study performed by Navarro et al. (1995) showing a 60MDa plasmid and a 37 MDa plasmid in freshwater and sediment Nitrobacter populations. In conjunction with Schutts’ (1990) study Navarro et al. (1995) illustrate genomic features that may play crucial roles in determining the distribution and ecological impact of Nitrobacter. Nitrifying bacteria in general tend to be less abundant than their heterotrophic counterparts, as the oxidizing reactions they perform have a low energy yield and most of their energy production goes toward carbon-fixation rather than growth and reproduction.
History
In 1890, Ukrainian-Russian microbiologist Sergei Winogradsky successfully isolated the first pure cultures of nitrifying bacteria which are capable of growth in the absence of organic matter and sunlight. The exclusion of organic material by Winogradsky in the preparation of his cultures is recognized as a contributing factor to his success in isolating the microbes. In 1891, English chemist Robert Warington proposed a two-stage mechanism for nitrification, mediated by two distinct genera of bacteria. The first stage is proposed as the conversion of ammonia to nitrite and the second the oxidation of nitrite to nitrate. Winogradsky named the bacteria responsible for the oxidation of nitrite to nitrate Nitrobacter in his subsequent study on microbial nitrification in 1892. Winslow et al. proposed the Type species Nitrobacter winogradsky in 1917. The species was officially recognized in 1980.
Main Species
Some sources regard Nitrobacteraceae to be the family of the genus Nitrobacter.
Species in the genus Nitrobacter include
