 | ||
Carboxysomes are bacterial organelles consisting of polyhedral protein shells filled with the enzyme Ribulose-1,5-biphosphate carboxylase/oxygenase (RuBisCO) -the predominant enzyme in carbon fixation and the rate limiting enzyme in the Calvin Cycle-and a carbonic anhydrase. Carboxysomes are thought to have evolved as a consequence of the increase in oxygen concentration in the ancient atmosphere; this is because oxygen is a competing substrate to carbon dioxide in the RuBisCO reaction. To overcome the inefficiency of RuBisCO, carboxysomes concentrate carbon dioxide inside the shell by means of co-localized carbonic anhydrase activity, which produces carbon dioxide from the bicarbonate that diffuses into the carboxysome. The resulting production of carbon dioxide near RuBisCO decreases the proportion of ribulose-1,5-bisphosphate oxygenation and thereby avoids costly photorespiratory reactions. The surrounding shell provides a barrier to carbon dioxide loss, helping to increase its concentration around RuBisCO. The carboxysome is an essential part of the carbon-concentrating mechanism (CCM).
Contents
- Discovery
- Architecture
- Two Types of Carboxysomes
- Alpha Carboxysomes
- Beta carboxysomes
- Potential uses of the carboxysome in biotechnology
- References
Carboxysomes are the best studied example of a bacterial microcompartment (BMC), the term for functionally diverse organelles that are alike in having a protein shell.
Discovery
Polyhedral bodies were discovered by transmission electron microscopy in the cyanobacterium Phormidium uncinatum in 1956. These were later observed in other cyanobacteria and in some chemotrophic bacteria that fixed carbon dioxide—many of them are sulfur reducers or nitrogen fixers (for example, Halothiobacillus, Acidithiobacillus, Nitrobacter and Nitrococcus). The polyhedral bodies were first purified from Thiobacillus neapolitanus (now Halothiobacillus neapolitanus) in 1973 and shown to contain RuBisCO, held within a rigid outer covering. The authors proposed that since these appeared to be organelles involved in carbon fixation, they should be called carboxysomes.
Architecture
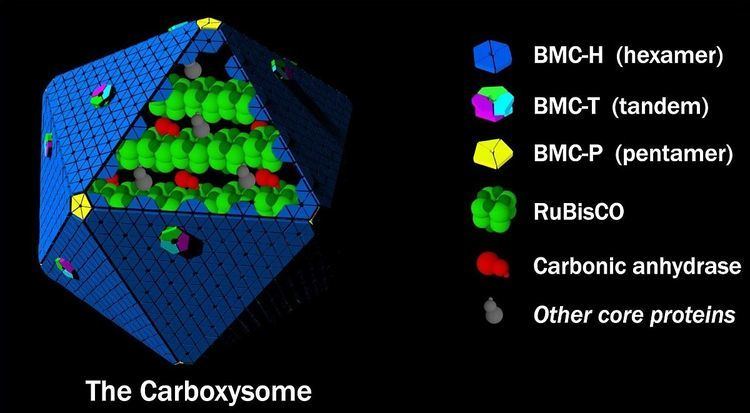
Structurally, carboxysomes are icosahedral, or quasi-icosahedral. Electron cryo-tomography studies have confirmed the approximately icosahedral geometry of the carboxysome, and have imaged protein molecules inside (presumed to be RuBisCO), arranged in a few concentric layers. The non-icosahedral faceted shapes of some carboxysomes can naturally be explained within the elastic theory of heterogeneous thin shells. The carboxysome has an outer shell composed of a few thousand protein subunits, which encapsulates a CO2-producing enzyme (carbonic anhydrase) and a carbon-fixing enzyme (RuBisCO). Proteins known to form the shell have been structurally characterized by X-ray crystallography. The protein that constitutes the majority of the shell forms a cyclical hexamer and belongs to BMC protein family. These hexamers, BMC-H proteins, are the basic building blocks of the shell. In some crystal forms the hexamers assemble further in a side-by-side fashion to form a tightly packed molecular layer, which presumably is how the facets of the shell are assembled. Small pores perforate many different types of BMC-H hexamers, and may serve as the route for diffusion of small substrates (e.g. bicarbonate) into and out of the carboxysome. Positively charged amino acids in the pores presumably help promote the diffusion of the negatively charged substrates and products. Other minor structural components of the shell that have been characterized include pentameric proteins (BMC-P proteins), which have been proposed to occupy the vertices of the icosahedral shell. A third building block of the carboxysome shell is a protein composed of two BMC domains in tandem (BMC-T proteins). Structurally, many of these are known to form trimers which are pseudohexameric. Some members of the BMC-T protein family stack in a face-to-face fashion and form tiny cages. Based on crystal structures, these protein cages have relatively large gated pores on both sides, and it has been proposed that the opening and closing of the pore could be controlled in a manner similar to an air-lock. Such an air-lock, in contrast to BMC-H proteins with constitutively open pores, has been suggested to serve as a route for larger substrates (ribulose-1,5-bisphosphate) and products (3-phosphoglycerate) that must cross the shell.
A number of viral capsids are also icosahedral, composed of hexameric and pentameric proteins, but currently there is no evidence suggesting any evolutionary relationship between the carboxysome shell and viral capsids.
Two Types of Carboxysomes
There are two types of carboxysomes. Although they may seem similar in appearance, they differ in their protein composition, including the form of RuBisCO they enclose. Furthermore, studies have revealed fundamental differences in their gene organization and possibly in how they assemble.
Alpha-Carboxysomes
Alpha-carboxysomes are also referred as the cso type of carboxysome. They contain Form IA RuBisCO; they are found in alpha-cyanobacteria, some nitrifying bacteria, some sulfur-oxidizing bacteria (for example, Halothiobacillus neapolitanus), and some purple bacteria. The alpha-carboxysome was the first bacterial microcompartment to be purified and characterized. Electron microscopy studies on purified alpha-carboxysomes or cell sections containing alpha-carboxysomes revealed that they are typically 100-160 nm in diameter. Common building blocks for the shell of alpha-carboxysomes are called CsoS1A/B/C (BMC-H), CsoS4A/B (BMC-P), and CsoS1D (BMC-T). CsoS4A/B were the first BMC-P proteins to be experimentally demonstrated as minor components of the BMC shell (only 12 pentamers are required to cap the vertices of an icosahedron). CsoS1D is first BMC-T which has been structurally characterized; it is also the first example of dimerization of two BMC building blocks in a face-to-face fashion to create a tiny cage. The CsoS1D cage has gated pore at both end, which is proposed to facilitate large metabolites crossing the shell. In addition to the specific form of RuBisCO, other encapsulated proteins distinguish alpha-carboxysomes from beta-carboxysomes such as CsoS2 and CsoSCA. The CsoS2 protein has a very high pI and a unique primary structure. The primary structure of CsoS2 appears tripartite, composed of an N-terminal, middle- and C-terminal regions. Repetitive motifs can be identified in the N-terminal and middle regions. Recently, it was proposed to be an intrinsically disordered protein with an essential role in alpha-carboxysome assembly. CsoSCA is a shell-associated beta-carbonic anhydrase. Studies in Halothiobacillus neapolitanus have shown that empty shells of normal shape and composition are assembled in carboxysomal RuBisCO-lacking mutants, suggesting that alpha-carboxysome shell biogenesis and enzyme sequestration are two independent, but functionally linked processes. Intriguingly, carboxysomes of Halothiobacillus neapolitanus have been found to accommodate chimeric and heterologous species of RuBisCO and it is the large subunit of RuBisCO which determines whether the enzyme is sequestered into carboxysomes or not.
Beta-carboxysomes
The signature proteins of the beta-carboxysome are Form IB RuBisCO and a gamma carbonic anhydrase homolog. Beta-carboxysomes are typically bigger than alpha-carboxysomes: the observed diameters for them vary from 200 to 400 nm. The structural proteins that are essential for carboxysome formation are encoded in the conserved carboxysome locus known as the ccm locus. The ccm locus includes genes for core proteins CcmM and CcmN and the shell proteins CcmK (a BMC-H protein), CcmL (a BMC-P protein) and CcmO (a BMC-T protein).
A full length CcmM protein consists of a gamma-carbonic anhydrase domain on and three to five small subunit-like domains (SSLDs; which resemble RbcS, the small subunit of RuBisCO) on its C-terminus. The ccmM gene contains an internal translation site that produces a short form of CcmM (a protein which only consists of SSLDs); both long and short forms of CcmM are required for carboxysome assembly. CcmN contains multiple hexapeptide-repeat domains on its N-terminus and a short α-helical encapsulation peptide on the C-terminus.
Other structural components of the carboxysomes are encoded outside of the ccm locus. CcmP is a BMC-T protein that is absolutely conserved among organisms that form beta-carboxysomes. CcmP pseudohexamer stacks to form a nanocompartment—an example of an air-lock forming protein. Likewise, in some cyanobacterial strains the beta-carboxysomes contain a beta-carbonic anhydrase that is not found in the ccm locus.
The beta-carboxysome assembles from the inside out, first an enzymatic core forms that is subsequently encapsulated by a protein shell. Carboxysome assembly occurs through a series of protein-protein interactions: the enzyme RuBisCO and the two isoforms (full length and short form) of the CcmM protein interact by means of the SSLDs; in strains containing CcaA the beta-carbonic anhydrase is brought into the carboxysome core by interaction with the N-terminus of the full length CcmM. Once the procarboxysome (the carboxysome core) is formed, the N-terminus of the adapter protein CcmN interacts with the N-terminus of CcmM, while the C-terminus of CcmN recruits the shell proteins CcmK (BMC-H) and CcmO (BMC-T). The final step is the addition of the vertices formed by the BMC-P protein CcmL, which then fully cap the enzymatic core.
Potential uses of the carboxysome in biotechnology
As the case with other BMCs, the carboxysome is attracting significant attention by researchers for applications in synthetic biology. The transfer of a genetic module coding for an alpha-carboxysome has been shown to produce carboxysome-like structures in E. coli. Bioengineering of carboxysome shells have been shown feasible and beta-carboxysomes constructed with chimeric proteins or with chimeric shells has been reported. If they can be installed in plants, carboxysomes may increase photosynthetic efficiency in plants. To this end, beta-carboxysomal proteins and Form IB RuBisCO have been expressed in tobacco but functional carboxysomes have not yet been obtained.
