Appearance white crystals | ||
 | ||
Niobium oxychloride is the inorganic compound with the formula NbOCl3. It is a white, crystalline, diamagnetic solid. It is often found as an impurity in samples of niobium pentachloride, a common reagent in niobium chemistry.
Contents
Structure
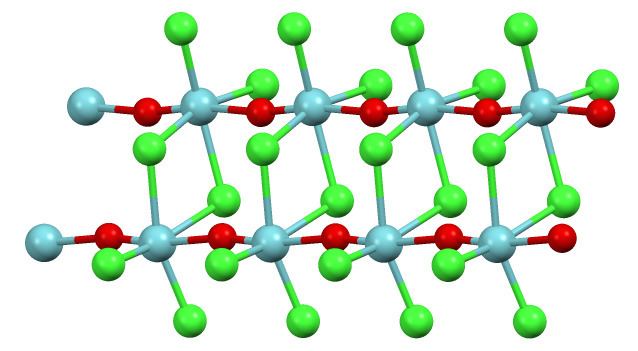
In the solid state the coordination sphere for niobium is a distorted octahedron. The Nb-O bonds and Nb-Cl bonds are unequal. This structure can be described as planar Nb2Cl6 core connected by O-Nb-O bridges. In this way, the compound is best described as a polymer, consisting of a double stranded chain.
In the gas phase above 320 °C the Raman spectrum is consistent with a pyramidal monomer containing a niobium oxygen double bond.
Synthesis
Niobium oxychloride is prepared by treating the pentachloride with oxygen:
NbCl5 + 1/2 O2 → NbOCl3 + Cl2This reaction is conducted at about 200 °C. NbOCl3 also forms as a major side-product in the reaction of niobium pentoxide with various chlorinating agents such as carbon tetrachloride and thionyl chloride.
2 Nb2O5 + 6 CCl4 → 4 NbOCl3 + 6 COCl2