Density 1.64 g/cm³ Boiling point 74.6 °C | Formula SOCl2 Molar mass 118.97 g/mol | |
 | ||
Appearance clear, colourless liquid (yellows on ageing) Related thionyl halides Related compounds | ||
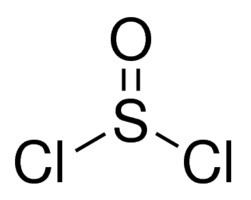
Socl2 lewis structure thionyl chloride
Thionyl chloride is an inorganic compound with the chemical formula SOCl2. It is a moderately volatile colourless liquid with an unpleasant acrid odour. Thionyl chloride is primarily used as a chlorinating reagent, with approximately 45,000 tons per year being produced during the early 1990s. It is toxic and will react violently with water to produce toxic gases, it is also listed as a Schedule 3 compound as it may be used for the production of chemical weapons.
Contents
- Socl2 lewis structure thionyl chloride
- Production
- Properties and structure
- Reactions
- With oxygen species
- With nitrogen species
- With sulphur species
- With phosphorus species
- With metals
- Other reactions
- Batteries
- Safety
- References
Thionyl chloride is sometimes confused with sulfuryl chloride, SO2Cl2, but the properties of these compounds differ significantly. Sulfuryl chloride is a source of chlorine whereas thionyl chloride is a source of chloride ions.
Production
The major industrial synthesis involves the reaction of sulfur trioxide and sulfur dichloride:
SO3 + SCl2 → SOCl2 + SO2Other methods include syntheses from phosphorus pentachloride, chlorine and sulfur dichloride, or phosgene:
SO2 + PCl5 → SOCl2 + POCl3SO2 + Cl2 + SCl2 → 2 SOCl2SO3 + Cl2 + 2 SCl2 → 3 SOCl2SO2 + COCl2 → SOCl2 + CO2The first of the above four reactions also affords phosphorus oxychloride (phosphoryl chloride), which resembles thionyl chloride in many of its reactions.
Properties and structure
SOCl2 adopts a pyramidal molecular geometry with Cs molecular symmetry. This geometry is attributed to the effects of the lone pairs on the sulfur(IV) centre.
In the solid state SOCl2 forms monoclinic crystals with the space group P21/c.
Stability
Thionyl chloride has a long shelf life, however "aged" samples develop a yellow hue, possibly due to the formation of disulfur dichloride. It slowly decomposes to S2Cl2, SO2 and Cl2 at just above the boiling point. Thionyl chloride is susceptible to photolysis, which primarily proceeds via a radical mechanism. Samples showing signs of ageing can be purified by distillation under reduced pressure, to give a clear colourless liquid.
Reactions
Thionyl chloride is mainly used in the industrial production of organochlorine compounds, which are often intermediates in pharmaceuticals and agrichemicals. It usually is preferred over other reagents, such as phosphorus pentachloride, as its by-products (HCl and SO2) are gaseous, which simplifies purification of the product.
Many of the products of thionyl chloride are themselves highly reactive and as such it is involved in a wide range of reactions.
With oxygen species
Thionyl chloride reacts with water to form sulfur dioxide and hydrochloric acid. This process is highly exothermic.
SOCl2 + H2O → 2 HCl + SO2Classically, it converts carboxylic acids to acyl chlorides.
SOCl2 + RCO2H → RC(O)Cl + SO2 + HClBy a similar process it also converts alcohols to alkyl chlorides. If the alcohol is chiral the reaction generally proceeds via an SNi mechanism with retention of stereochemistry; however, depending on the exact conditions employed, stereo-inversion can also be achieved. Historically the use of SOCl2 in combination with a tertiary amine such as pyridine was called the Darzens halogenation however this name is rarely used by modern chemists.
Reactions with an excess of alcohol produce sulfite esters, which can be powerful methylation, alkylation and hydroxyalkylation reagents.
SOCl2 + 2 ROH → (RO)2SO + 2 HClFor example, the addition of SOCl2 to amino acids in methanol selectively yields the corresponding methyl esters.
With nitrogen species
With primary amines, thionyl chloride gives the sulfinylamine derivatives (RNSO), one example being N-sulfinylaniline. Thionyl chloride reacts with primary formamides to form isocyanides and with secondary formamides to give chloroiminium ions; as such a reaction with dimethylformamide will form the Vilsmeier reagent. By an analogous process primary amides will react with thionyl chloride to form imidoyl chlorides, with secondary amides also giving chloroiminium ions. These species are highly reactive and can be used to catalyse the conversion of carboxylic acids to acyl chlorides, they are also exploited in the Bischler–Napieralski reaction as a means of forming isoquinolines.
Primary amides will continue on to form nitriles if heated (Von Braun amide degradation). Thionyl chloride has also been used to promote the Beckmann rearrangement of oximes.
With sulphur species
With phosphorus species
Thionyl chloride converts phosphonic acids and phosphonates into phosphoryl chlorides. It is for this type of reaction that thionyl chloride is listed as a Schedule 3 compound, as it can be used in the "di-di" method of producing G-series nerve agents. For example, thionyl chloride converts dimethyl methylphosphonate into methylphosphonic acid dichloride, which can be used in the production of sarin and soman.
With metals
As SOCl2 reacts vigorously with water it can be used to dehydrate various metal chloride hydrates, for example MgCl2⋅6H2O, AlCl3⋅6H2O, FeCl3⋅6H2O etc. This conversion involves treatment with refluxing thionyl chloride and follows the following general equation:
MCln·xH2O + x SOCl2 → MCln + x SO2 + 2x HClOther reactions
Batteries
Thionyl chloride is a component of lithium-thionyl chloride batteries, where it acts as the positive electrode (cathode) with lithium forming the negative electrode (anode); the electrolyte is typically lithium tetrachloroaluminate. The overall discharge reaction is as follows:
4Li + 2 SOCl2 → 4 LiCl + S + SO2These non rechargeable batteries have many advantages over other forms of lithium battery such as a high energy density, a wide operational temperature range and long storage and operational lifespans. However, their high cost and safety concerns have limited their use. The contents of the batteries are highly toxic and require special disposal procedures, additionally they may explode if shorted.
Safety
SOCl2 is a reactive compound that can violently and/or explosively release dangerous gases upon contact with water and other reagents. Thionyl chloride is controlled under the Chemical Weapons Convention, where it is listed in Schedule 3. Thionyl chloride is used in the "di-di" method of producing G-series nerve agents.
