Pregnancycategory C (USA) ATC code C01DA14 (WHO) Protein binding <5% Molar mass 191.139 g/mol | Routes ofadministration oral Bioavailability >95% Formula C6H9NO6 CAS ID 16051-77-7 | |
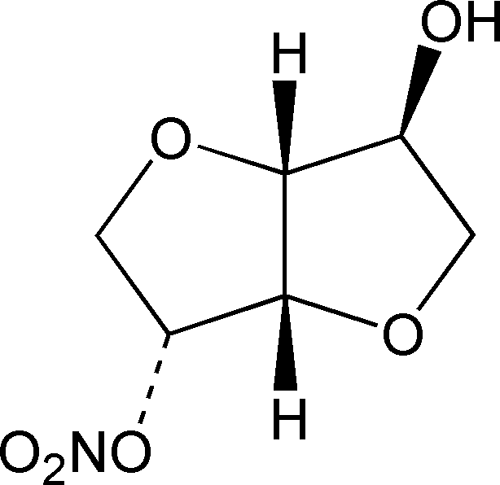 | ||
AHFS/Drugs.com Consumer Drug Information IUPAC ID 8-nitrooxy-2,6-dioxabicyclo[3.3.0]octan-4-ol | ||
Isosorbide mononitrate is a drug used principally in the treatment of angina pectoris and acts by dilating the blood vessels so as to reduce the blood pressure. It is sold in the USA by Kremers Urban under the trade name Monoket, also sold in the USA under the name Imdur, and marketed in the UK under the trade names: Monosorb, Chemydur. In India, this drug is available under the brand names of Ismo, Isonorm, Monotrate, Solotrate, and Monit. In Russia it is occasionally used under the brand names Monocinque and Pektrol.
Contents
182 isosorbide dinitrate or isosorbide mononitrate
Uses
Isosorbide mononitrate is a nitrate-class drug used for the prophylactic treatment of angina pectoris; that is, it is taken in order to prevent or at least reduce the occurrence of angina. Research on isosorbide mononitrate as a cervical ripener to reduce time at hospital to birth is supportive.
Side-effects
The adverse reactions, which follow, have been reported in studies with isosorbide mononitrate:
Very common: Headache predominates (up to 30%) necessitating withdrawal of 2 to 3% of patients, but the incidence reduces rapidly as treatment continues .
Common: Tiredness, sleep disturbances (6%) and gastrointestinal disturbances (6%) have been reported during clinical trials with isosorbide mononitrate modified-release tablets, but at a frequency no greater than for placebo. Hypotension (4 to 5%), poor appetite (2.5%), nausea (1%).
Adverse effects associated with the clinical use of the drug are as expected with all nitrate preparations. They occur mainly in the early stages of treatment.
Hypotension (4%) with symptoms such as dizziness and nausea (1%) have been reported. In general, these symptoms disappear during long-term treatment.
Other reactions that have been reported with isosorbide mononitrate modified-release tablets include tachycardia, vomiting, diarrhoea, vertigo, and heartburn.
