Symbol HATPase_c Pfam clan CL0025 SMART SM00387 | Pfam PF02518 InterPro IPR003594 SCOP 1ei1 | |
 | ||
Hsp90 (heat shock protein 90) is a chaperone protein that assists other proteins to fold properly, stabilizes proteins against heat stress, and aids in protein degradation. It also stabilizes a number of proteins required for tumor growth, which is why Hsp90 inhibitors are investigated as anti-cancer drugs.
Contents
- Isoforms
- Common features
- Domain structure
- N terminal domain
- Middle domain
- C terminal domain
- Mechanism
- ATP binding
- Protein binding
- Normal cells
- Protein folding and role as chaperone
- Protein degradation
- Interaction with steroid receptors
- Cancerous cells
- Clinical significance
- Evolution
- References
Heat shock proteins, as a class, are among the most highly expressed cellular proteins across all species. As their name implies, heat shock proteins protect cells when stressed by elevated temperatures. They account for 1–2% of total protein in unstressed cells. However, when cells are heated, the fraction of heat shock proteins increases to 4–6% of cellular proteins.
Heat shock protein 90 (Hsp90) is one of the most common of the heat-related proteins. The "90" comes from the fact that it weighs roughly 90 kiloDaltons. A 90 kDa protein is considered fairly large for a non-fibrous protein. Hsp90 is found in bacteria and all branches of eukarya, but it is apparently absent in archaea. Whereas cytoplasmic Hsp90 is essential for viability under all conditions in eukaryotes, the bacterial homologue HtpG is dispensable under non-heat stress conditions.
This protein was first isolated by extracting proteins from cells stressed by heating, dehydrating or by other means, all of which caused the cell’s proteins to begin to denature. However it was later discovered that Hsp90 also has essential functions in unstressed cells.
Isoforms
Hsp90 is highly conserved and expressed in a variety of different organisms from bacteria to mammals – including the prokaryotic analogue HtpG (high-temperature protein G) with 40% sequence identity and 55% similarity to the human protein. Yeast Hsp90 is 60% identical to human Hsp90α.
In mammalian cells, there are two or more genes encoding cytosolic Hsp90 homologues, with the human Hsp90α showing 85% sequence identity to Hsp90β. The α- and the β-forms are thought to be the result of a gene duplication event that occurred millions of years ago.
The five functional human genes encoding Hsp90 protein isoforms are listed below:
There are 12 human pseudogenes (non-functional genes) that encode additional Hsp90 isoforms that are not expressed as proteins.
A membrane-associated variant of cytosolic Hsp90, lacking an ATP-binding site, has recently been identified and was named Hsp90N. This HSP90α-Δ-N transcript is a chimera, with the first 105 bp of the coding sequence derived from the CD47 gene on chromosome 3q13.2, and the remaining coding sequence derived from HSP90AA1. However, gene-encoding Hsp90N was later proven to be non-existent in human genome. It is possibly a cloning artifact or a product of chromosomal rearrangement occurring in a single cell line.
Common features
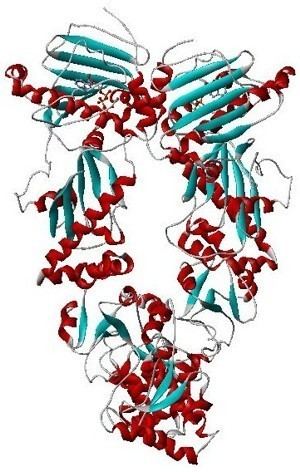
The overall structure of Hsp90 is similar to that of other proteins in that it contains all of the common secondary structural elements (i.e., alpha helixes, beta pleated sheets, and random coils). Being a cytoplasmic protein requires that the protein be globular in structure, that is largely non-polar on the inside and polar on the outside, so as to be dissolved by water. Hsp90 contains nine helices and eight anti-parallel beta pleated sheets, which combine to form several alpha/beta sandwiches. The 310 helices make up approximately 11% of the protein's amino acid residues, which is much higher than the average 4% in other proteins.
Domain structure
Hsp90 consists of four structural domains:
Crystal structures are available for the N-terminal domain of yeast and human Hsp90, for complexes of the N-terminus with inhibitors and nucleotides, and for the middle domain of yeast Hsp90. Recently structures for full length Hsp90 from E. coli (2IOP, 2IOQ), yeast (2CG9, 2CGE), and the dog endoplasmic reticulum (2O1U, 2O1V) were elucidated.
Hsp90 forms homodimers where the contact sites are localized within the C-terminus in the open conformation of the dimer. The N-termini also come in contact in the closed conformation of the dimer.
N-terminal domain
The N-terminal domain shows homology not only among members of the Hsp90 chaperone family but also to members of the ATPase/kinase GHKL (Gyrase, Hsp90, Histidine Kinase, MutL) superfamily.
A common binding pocket for ATP and the inhibitor geldanamycin is situated in the N-terminal domain. Amino acids that are directly involved in the interaction with ATP are Leu34, Asn37, Asp79, Asn92, Lys98, Gly121, and Phe124. In addition, Mg2+ and several water molecules form bridging electrostatic and hydrogen bonding interactions, respectively, between Hsp90 and ATP. In addition, Glu33 is required for ATP hydrolysis.
Middle domain
The middle domain is divided into three regions:
The MD is also involved in client protein binding. For example, proteins known to interact this the Hsp90 MD include PKB/Akt1, eNOS, Aha1, Hch1. Furthermore, substrate binding (e.g., by Aha1 and Hch1) to the MD is also known to increase the ATPase activity of Hsp90.
C-terminal domain
The C-terminal domain possesses an alternative ATP-binding site, which becomes accessible when the N-terminal Bergerat pocket is occupied.
At the very C-terminal end of the protein is the tetratricopeptide repeat (TPR) motif recognition site, the conserved MEEVD pentapeptide, that is responsible for the interaction with co-factors such as the immunophilins FKBP51 and FKBP52, the stress induced phosphoprotein 1 (Sti1/Hop), cyclophilin-40, PP5, Tom70, and many more.
Mechanism
The Hsp90 protein contains three functional domains, the ATP-binding, protein-binding, and dimerizing domain, each of which playing a crucial role in the function of the protein.
ATP binding
The region of the protein near the N-terminus has a high-affinity ATP-binding site. The ATP binds to a sizable cleft in the side of protein, which is 15 Å (1.5 nanometres) deep. This cleft has a high affinity for ATP, and when given a suitable protein substrate, Hsp90 cleaves the ATP into ADP and Pi. Direct inhibitors of ATP binding or allosteric inhibitors of either ATP binding or ATPase activity can block Hsp90 function. Another interesting feature of the ATP-binding region of Hsp90 is that it has a “lid” that is open during the ADP-bound state and closed in the ATP-bound state, in the open conformation, the lid has no intraprotein interaction, and when closed comes into contact with several residues. The contribution of this lid to the activity of Hsp90 has been probed with site-directed mutagenesis. The Ala107Asp mutant stabilizing the closed conformation of the protein through the formation of additional hydrogen bonds substantially increases ATPase activity while leaving the AMP+PnP conformation unchanged.
The ATPase-binding region of Hsp90 is currently under intense study, because it is the principal binding site of drugs targeting this protein. Antitumor drugs targeting this section of Hsp90 include the antibiotics geldanamycin, herbimycin, radicicol, deguelin, derrubone, macbecin, and beta-lactams.
Protein binding
The protein-binding region of Hsp90 is located toward the C-terminus of the amino sequence. The Hsp90 protein can adopt two major conformational states. The first is an open ATP-bound state and the second is a closed ADP-bound state. Thus, ATP hydrolysis drives what is commonly referred to as a “pincer-type” conformational change in the protein binding site.
Hsp90, while in the open conformation, leaves some hydrophobic residues exposed, to which unfolded and misfolded proteins that have unusual hydrophobic regions exposed are recruited with high affinity. When a bound substrate is in place, the energy-releasing ATP hydrolysis by the ATPase function near the N-terminal domain forces conformational changes that clamp the Hsp90 down onto the substrate. In a reaction similar to that of other molecular clamp proteins like GyrB and MutL, this site drives virtually all of the protein folding functions that Hsp90 plays a role in. In contrast, MutL and GyrB function as topoisomerases and use a charge clamp with a high amount of positively charged sidechains that is electrostatically attracted to the negative backbone of DNA.
The ability of Hsp90 to clamp onto proteins allows it to perform several functions including assisting folding, preventing aggregation, and facilitating transport.
Normal cells
In unstressed cells, Hsp90 plays a number of important roles, which include assisting folding, intracellular transport, maintenance, and degradation of proteins as well as facilitating cell signaling.
Protein folding and role as chaperone
Hsp90 is known to associate with the non-native structures of many proteins, which has led to the proposal that Hsp90 is involved in protein folding in general. Furthermore, Hsp90 has been shown to suppress the aggregation of a wide range of "client" or "substrate" proteins and hence acts as a general protective chaperone. However Hsp90 is somewhat more selective than other chaperones.
Protein degradation
Eukaryotic proteins that are no longer needed or are misfolded or otherwise damaged are usually marked for destruction by the polyubiquitation pathway. These ubiquitinated proteins are recognized and degraded by the 26S proteasome. Hence the 26S proteasome is an integral part of the cell's mechanism to degrade proteins. Furthermore, a constant supply of functional Hsp90 is needed to maintain the tertiary structure of the proteasome. Finally experiments done with heat sensitive Hsp90 mutants and the 26S proteasome suggest that Hsp90 is responsible for most, if not all, of the ATPase activity of the proteasome.
Interaction with steroid receptors
The glucocorticoid receptor (GR) is the most thoroughly studied example of a steroid receptor whose function is crucially dependent on interactions with Hsp90. In the absence of the steroid hormone cortisol, GR resides in the cytosol complexed with several chaperone proteins including Hsp90 (see figure to the right). These chaperones maintain the GR in a state capable of binding hormone. A second role of Hsp90 is to bind immunophilins (e.g., FKBP52) that attach the GR complex to the dynein protein trafficking pathway, which translocates the activated receptor from the cytoplasm into the nucleus. Once in the nucleus, the GR dimerizes and binds to specific sequences of DNA and thereby upregulates the expression of GR responsive genes. Hsp90 is also required for the proper functioning of several other steroid receptors, including those responsible for the binding of aldosterone, androgen, estrogen, and progesterone.
Cancerous cells
Cancerous cells overexpress a number of proteins, including growth factor receptors, such as EGFR, or signal transduction proteins such as PI3K and AKT (Inhibition of these proteins may trigger apoptosis). Hsp90 stabilizes various growth factor receptors and some signaling molecules including PI3K and AKT proteins. Hence inhibition of Hsp90 may induce apoptosis through inhibition of the PI3K/AKT signaling pathway and growth factor signaling generally.
Another important role of Hsp90 in cancer is the stabilization of mutant proteins such as v-Src, the fusion oncogene Bcr/Abl, and mutant forms of p53 that appear during cell transformation. It appears that Hsp90 can act as a "protector" of less stable proteins produced by DNA mutations.
Hsp90 is also required for induction of vascular endothelial growth factor (VEGF) and nitric oxide synthase (NOS). Both are important for de novo angiogenesis that is required for tumour growth beyond the limit of diffusion distance of oxygen in tissues. It also promotes the invasion step of metastasis by assisting the matrix metalloproteinase MMP2. Together with its co-chaperones, Hsp90 modulates tumour cell apoptosis "mediated through effects on AKT, tumor necrosis factor receptors (TNFR) and nuclear factor-κB (NF-κB) function." Also, Hsp90 participates in many key processes in oncogenesis such as self-sufficiency in growth signals, stabilization of mutant proteins, angiogenesis, and metastasis.
Clinical significance
Hsp90 plays apparently conflicting roles in the cell, as it is essential for both the creation and the maintenance as well as the destruction of proteins. Its normal function is critical to maintaining the health of cells, whereas its dysregulation may contribute to carcinogenesis. The ability of this chaperone to both stabilize the 26S proteasome (which enables the cell to degrade unwanted and/or harmful proteins) and to stabilize kinases against the same proteasome demonstrates its functional diversity. The uses of Hsp90 inhibitors in cancer treatment highlight Hsp90's importance as a therapeutic target.
Targeting Hsp90 with drugs has shown promising effects in clinical trials. For example, the Hsp90 inhibitor geldanamycin has been used as an anti-tumor agent. The drug was originally thought to function as a kinase inhibitor but was subsequently shown to be an Hsp90 inhibitor where it uses a compact conformation to insert itself into the ATP binding site.
HSP90 beta has been identified as one of the autoantigenic biomarkers and targets involved in human ovarian autoimmune disease leading to ovarian failure and thereby infertility.
Prediction and validation of the immunodominant epitope/s of HSP90 beta protein has been demonstrated using sera from infertile women having anti-HSP90 autoantibodies. The decapeptide EP6 (380-389)is a major immunogenic epitope of HSP90 followed by EP1 (1-12) and EP8 (488-498). Knowledge of binding epitopes on the autoantigen is necessary to understand the subsequent pathologic events. Predicted 3D structures of these peptides demonstrated that they exist in the loop conformation, which is the most mobile part of the protein. Also, analysis of the sequences of HSP90 beta across several species reveals that EP6 peptide forms a part of a well-conserved motif. A polyclonal antibody generated to the immunodominant epitope- EP6 confirms similar biochemical and cellular immunoreactivity as seen with the patients' sera with anti-HSP90 autoantibodies. The study might generate new tools for the detection of disease-inducing epitopes and a possible therapeutic intervention.
Evolution
Sequence alignments of Hsp90 have shown the protein to have about 40% sequence identity across all homologs, indicating that it is a highly conserved protein. There are two homologs, found in the cytosol and endoplasmic reticulum respectively. The presence of these two homologs was likely caused by a gene duplication event very early in the evolution of eukaryotes that may have accompanied the evolution of the endoplasmic reticulum or the nucleus. This inference is supported by the fact that the duplication is found in Giardia lamblia, one of the earliest branching eukaryotic species. At least 2 other subsequent gene duplications occurred, which explains the different forms of Hsp90 found in fungi and vertebrates. One divergence produced cognate and heat-induced forms of Hsp90 in Saccharomyces cerevisiae, while the second gene duplication event in the cytosolic branch produced the alpha and beta subfamilies of sequences that are found in all vertebrates. In a phylogenetic tree based on Hsp90 sequences, it was found that plants and animals are more closely related to each other than to fungi. Similar to the Hsp90 protein, the gene for Hsp70 protein also underwent duplication at a very early stage in the formation of eukaryotic cells and the homologs in the cytosol and endoplasmic reticulum resulted from this gene duplication event. These gene duplication events are important in terms of the origin of the eukaryotic cell and of the endoplasmic reticulum.
