Specialty rheumatology ICD-9-CM 446.21 DiseasesDB 5363 | ICD-10 M31.0 (ILDS M31.010) OMIM 233450 MedlinePlus 000142 | |
 | ||
Goodpasture syndrome (GPS; also known as Goodpasture’s disease, antiglomerular basement antibody disease, or anti-GBM disease) is a rare autoimmune disease in which antibodies attack the basement membrane in lungs and kidneys, leading to bleeding from the lungs and kidney failure. It is thought to attack the alpha-3 subunit of type IV collagen, which has therefore been referred to as Goodpasture's antigen. Goodpasture syndrome may quickly result in permanent lung and kidney damage, often leading to death. It is treated with immunosuppressant drugs such as corticosteroids and cyclophosphamide, and with plasmapheresis, in which the antibodies are removed from the blood.
Contents
The disease was first described by an American pathologist Ernest Goodpasture of Vanderbilt University, in 1919 and was later named in his honor.
Signs and symptoms
The antiglomerular basement membrane (GBM) antibodies primarily attack the kidneys and lungs, although, generalized symptoms like malaise, weight loss, fatigue, fever, and chills are also common, as are joint aches and pains. 60 to 80% of those with the condition experience both lung and kidney involvement; 20-40% have kidney involvement alone, and less than 10% have lung involvement alone. Lung symptoms usually antedate kidney symptoms and usually include: coughing up blood, chest pain (in less than 50% of cases overall), cough, and shortness of breath. Kidney symptoms usually include blood in the urine, protein in the urine, unexplained swelling of limbs or face, high amounts of urea in the blood, and high blood pressure.
Cause
Its precise cause is unknown, but an insult to the blood vessels taking blood from and to the lungs is believed to be required to allow the anti-GBM antibodies to come into contact with the alveoli. Examples of such an insult include:
Pathophysiology
GPS causes the abnormal production of anti-GBM antibodies, by the plasma cells of the blood. The anti-GBM antibodies attack the alveoli and glomeruli basement membranes. These antibodies bind their reactive epitopes to the basement membranes and activate the complement cascade, leading to the death of tagged cells. T cells are also implicated. It is generally considered a type II hypersensitivity reaction.
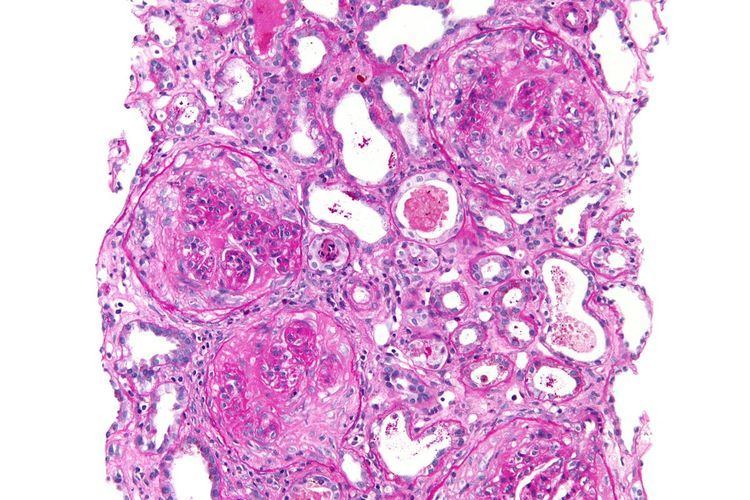
Diagnosis
The diagnosis of GPS is often difficult, as numerous other diseases can cause the various manifestations of the condition and the condition itself is rare. The most accurate means of achieving the diagnosis is testing the affected tissues by means of a biopsy, especially the kidney, as it is the best-studied organ for obtaining a sample for the presence of anti-GBM antibodies. On top of the anti-GBM antibodies implicated in the disease, about one in three of those affected also has cytoplasmic antineutrophilic antibodies in their bloodstream, which often predates the anti-GBM antibodies by about a few months or even years. The later the disease is diagnosed, the worse the outcome is for the affected person.
Treatment
The major mainstay of treatment for GPS is plasmapheresis, a procedure in which the affected person's blood is sent through a centrifuge and the various components separated based on weight. The plasma, clear liquid part of the blood, contains the anti-GBM antibodies that attacks the affected person's lungs and kidneys and is filtered out. The other parts of the blood, that is, the red blood cells, white blood cells, and platelets, are recycled and given intravenously as a replacement fluid. Most individuals affected by the disease also need to be treated with immunosuppressant drugs, especially cyclophosphamide, prednisone, and rituximab, to prevent the formation of new anti-GBM antibodies so as to prevent further damage to the kidneys and lungs. Other, less toxic immunosuppressants such as azathioprine may be used to maintain remission.
Prognosis
With treatment the five-year survival rate is >80% and fewer than 30% of affected individuals require long-term dialysis. A study performed in Australia and New Zealand demonstrated that in patients requiring renal replacement therapy (including dialysis) the median survival time is 5.93 years. Without treatment, virtually every affected person will end up dying from either advanced kidney failure or lung hemorrhages.
Epidemiology
GPS is rare, affecting about 0.5-1.8 per million people per year in Europe and Asia. It is also unusual among autoimmune diseases in that it is more common in males than in females and is also less common in blacks than whites, but more common in the Māori people of New Zealand. The peak age ranges for the onset of the disease are 20-30 and 60–70 years.
