Symbol FBN1 Entrez 2200 OMIM 134797 | Alt. symbols FBN, MFS1, WMS HUGO 3603 PDB 2W86 | |
 | ||
Marfan s syndrome fibrillin defect
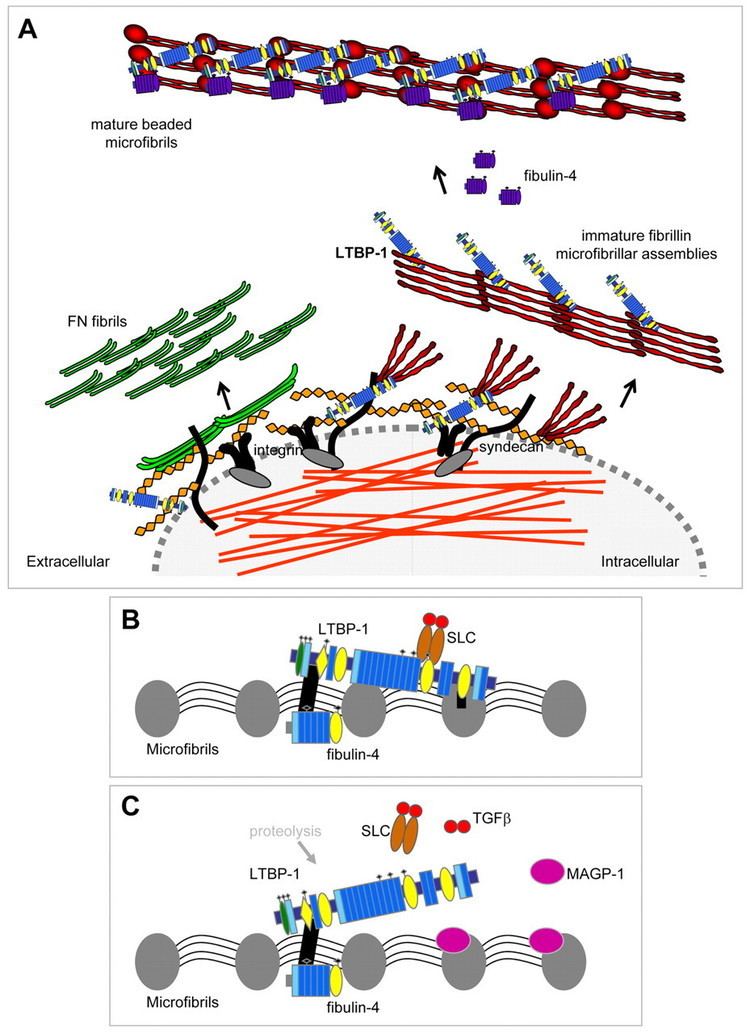
Fibrillin is a glycoprotein, which is essential for the formation of elastic fibers found in connective tissue. Fibrillin is secreted into the extracellular matrix by fibroblasts and becomes incorporated into the insoluble microfibrils, which appear to provide a scaffold for deposition of elastin.
Contents
- Marfan s syndrome fibrillin defect
- Osteogenesis imperfecta fibrillin and elastin
- Clinical aspect
- Fibrillin 1
- Structure
- Fibrillin 2
- Fibrillin 3
- Fibrillin 4
- References
Osteogenesis imperfecta fibrillin and elastin
Clinical aspect
Mutations in FBN1 and FBN2 are associated with adolescent idiopathic scoliosis .
Fibrillin-1
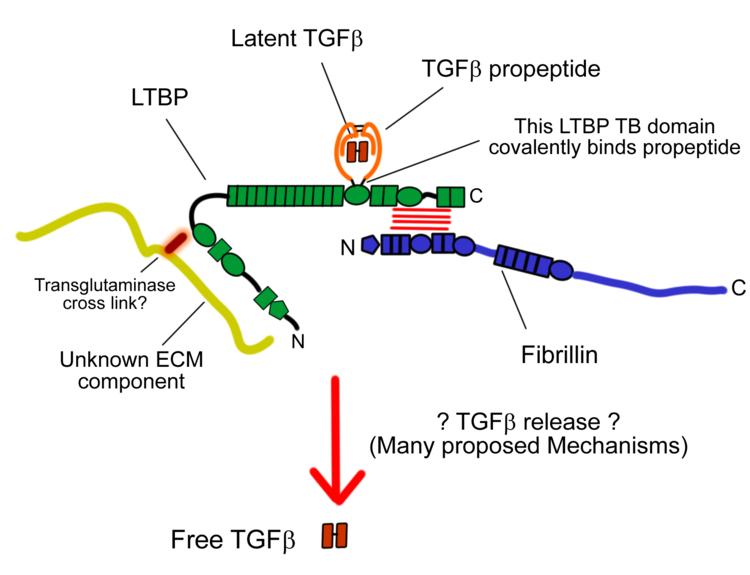
Fibrillin-1 is a major component of the microfibrils that form a sheath surrounding the amorphous elastin. It is believed that the microfibrils are composed of end-to-end polymers of fibrillin. To date, 3 forms of fibrillin have been described. The fibrillin-1 protein was isolated by Engvall in 1986, and mutations in the FBN1 gene cause Marfan syndrome.
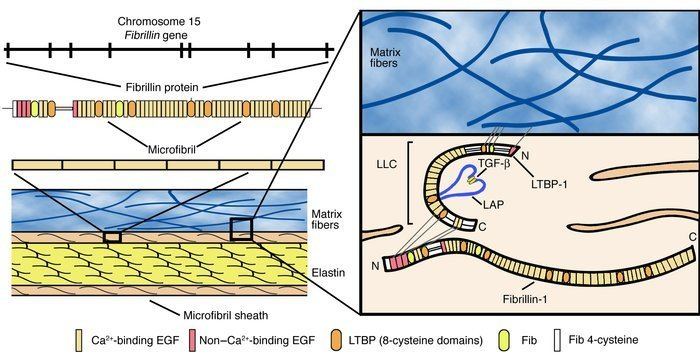
This protein is found in humans, and its gene is found on chromosome 15. At present more than 1500 different mutations have been described.
Structure
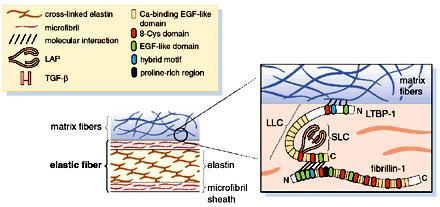
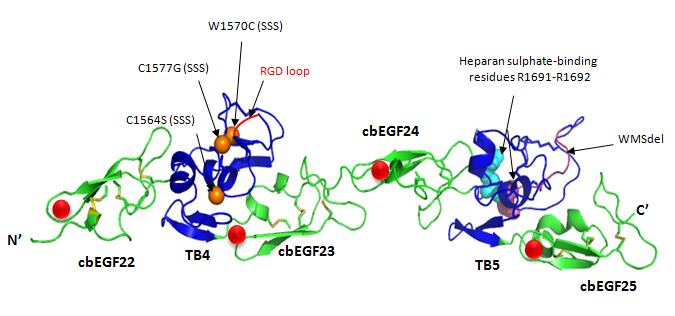
There is no complete, high-resolution structure of fibrillin-1. Instead, short fragments have been produced recombinantly and their structures solved by X-ray crystallography or using NMR spectroscopy. A recent example is the structure of the fibrillin-1 hybrid2 domain, in context of its flanking calcium binding epidermal growth factor domains, which was determined using X-ray crystallography to a resolution of 1.8 Å. The microfibrils that are made up of fibrillin protein are responsible for different cell-matrix interactions in the human body.
Fibrillin-2
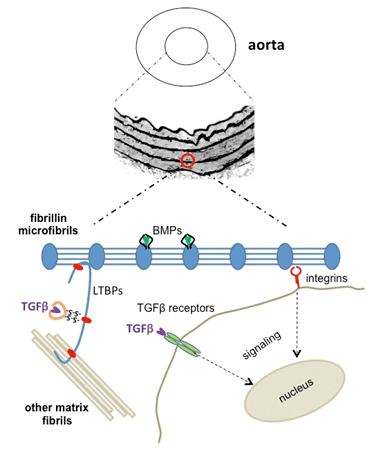
Fibrillin-2 was isolated in 1994 by Zhang and is thought to play a role in early elastogenesis. Mutations in the fibrillin-2 gene have been linked to Beal's Syndrome.
Fibrillin-3
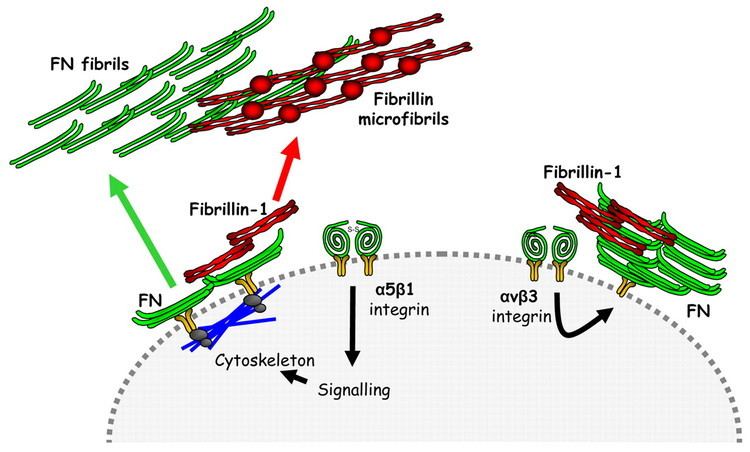
More recently, fibrillin-3 was described and is believed to be located mainly in the brain. Along with the brain, fibrillin-3 has been localized in the gonads and ovaries of field mice.
Fibrillin-4
Fibrillin-4 was first discovered in zebrafish, and has a sequence similar to fibrillin-2.
