 | ||
Anthocyanins (also anthocyans; from Greek: ἀνθός (anthos) = flower + κυανός (kyanos) = blue) are water-soluble vacuolar pigments that may appear red, purple, or blue depending on the pH. They belong to a parent class of molecules called flavonoids synthesized via the phenylpropanoid pathway; they are odorless but flavorful, contributing to taste as a moderately astringent sensation. Anthocyanins occur in all tissues of higher plants, including leaves, stems, roots, flowers, and fruits. Anthoxanthins are clear, white to yellow counterparts of anthocyanins occurring in plants. Anthocyanins are derived from anthocyanidins by adding sugars.
Contents
- Flavonoids anthocyanins red purple blue pigments
- Coloration
- Physiological roles
- Potential food value
- Light absorbance
- Occurrence of anthocyanins
- In flowers
- In food
- In leaves of plant foods
- Effect on autumn leaf color
- Flavylium cation derivatives
- Glycosides of anthocyanidins
- Stability
- pH
- Use as environmental pH indicator
- Biosynthesis
- Genetic analysis
- Dye sensitized solar cells
- Research
- Use as visual markers to mark genetically modified materials
- References
Flavonoids anthocyanins red purple blue pigments
Coloration
In flowers, the coloration that is provided by anthocyanin accumulation can attract a wide variety of animal pollinators, while in fruits, the same coloration can aid in seed dispersal by attracting herbivorous animals to the potentially edible fruits bearing these red, blue, or purple colors.
Physiological roles
Anthocyanins have an antioxidant role in plants against reactive oxygen species caused by abiotic stresses, such as overexposure to ultraviolet light and extreme temperatures. Tomato plants protect against cold stress with anthocyanins countering reactive oxygen species, leading to a lower rate of cell death in leaves.
Potential food value
Anthocyanins are considered secondary metabolites as a food additive with E number E163 (INS number 163); they are approved for use as a food additive in the EU, Australia and New Zealand.
Although anthocyanins have antioxidant properties in vitro, this antioxidant effect is not conserved after the plant is consumed. As interpreted by the Linus Pauling Institute and European Food Safety Authority, dietary anthocyanins and other flavonoids have little or no direct antioxidant food value following digestion. Unlike controlled test-tube conditions, the fate of anthocyanins in vivo shows they are poorly conserved (less than 5%), with most of what is absorbed existing as chemically modified metabolites that are rapidly excreted.
The increase in antioxidant capacity of blood seen after the consumption of anthocyanin-rich foods may not be caused directly by the anthocyanins, but instead may result from increased uric acid levels derived from metabolism of flavonoids.
Light absorbance
The absorbance pattern responsible for the red color of anthocyanins may be complementary to that of green chlorophyll in photosynthetically active tissues such as young Quercus coccifera leaves. It may protect the leaves from attacks by plant eaters that may be attracted by green color.
Occurrence of anthocyanins
Anthocyanins are found in the cell vacuole, mostly in flowers and fruits but also in leaves, stems, and roots. In these parts, they are found predominantly in outer cell layers such as the epidermis and peripheral mesophyll cells.
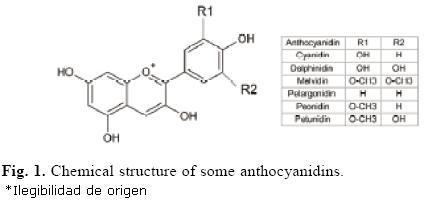
Most frequently occurring in nature are the glycosides of cyanidin, delphinidin, malvidin, pelargonidin, peonidin, and petunidin. Roughly 2% of all hydrocarbons fixed in photosynthesis are converted into flavonoids and their derivatives such as the anthocyanins. Not all land plants contain anthocyanin; in the Caryophyllales (including cactus, beets, and amaranth), they are replaced by betalains. Anthocyanins and betalains have never been found in the same plant.
Sometimes bred purposely for high anthocyanin quantities, ornamental plants such as sweet peppers may have unusual culinary and aesthetic appeal.
In flowers
Anthocyanins occur in the flowers of many plants, such as the famously blue poppies of some Meconopsis species and cultivars.
In food
Plants rich in anthocyanins are Vaccinium species, such as blueberry, cranberry, and bilberry; Rubus berries, including black raspberry, red Raspberry, and blackberry; blackcurrant, cherry, eggplant (aubergine) peel, black rice, Concord grape, muscadine grape, red cabbage, and violet petals. Red-fleshed peaches and apples contain anthocyanins. Anthocyanins are less abundant in banana, asparagus, pea, fennel, pear, and potato, and may be totally absent in certain cultivars of green gooseberries.
The highest recorded amount appears to be specifically in the seed coat of black soybean (Glycine max L. Merr.) containing around 2 g per 100 g, in purple corn kernels and husks, and in skins and pulp of black chokeberry (Aronia melanocarpa L.) (see table). Due to critical differences in sample origin, preparation and extraction methods determining anthocyanin content, the values presented in the adjoining table are not directly comparable.
Nature, traditional agriculture, and plant breeding have produced various uncommon crops containing anthocyanins, including blue- or red-flesh potatoes and purple or red broccoli, cabbage, cauliflower, carrots, and corn. Garden tomatoes have been subjected to a breeding program using introgression lines of genetically modified organisms (but not incorporating them in the final purple tomato) to define the genetic basis of purple coloration in wild species originally from Chile and the Galapagos Islands. The variety known as "Indigo Rose" became commercially available to the agricultural industry and home gardeners in 2012. Investing tomatoes with high anthocyanin content doubles their shelf-life and inhibits growth of a post-harvest mold pathogen, Botrytis cinerea.
Tomatoes have also been genetically modified with transcription factors from snapdragons to produce high levels of anthocyanins in the fruits. Anthocyanins can also be found in naturally ripened olives, and are partly responsible for the red and purple colors of some olives.
In leaves of plant foods
Content of anthocyanins in the leaves of colorful plant foods, such as purple corn, blueberries or lingonberries, is about 10 times higher than in the edible kernels or fruit.
The color spectrum of grape berry leaves can be analysed to evaluate the amount of anthocianins. Fruit maturity, quality and harvest time can be evaluated on the basis of the spectrum analysis.
Effect on autumn leaf color
The reds, purples, and their blended combinations responsible for autumn foliage come from anthocyanins. Unlike carotenoids, anthocyanins are not present in the leaf throughout the growing season, but are actively produced towards the end of summer. They develop in late summer in the sap of leaf cells, resulting from complex interactions of factors inside and outside the plant. Their formation depends on the breakdown of sugars in the presence of light as the level of phosphate in the leaf is reduced.
Anthocyanins are present in about 10% of tree species in temperate regions, although in certain areas such as New England, up to 70% of tree species may produce anthocyanins.
Flavylium cation derivatives
See Anthocyanidins article.
Glycosides of anthocyanidins
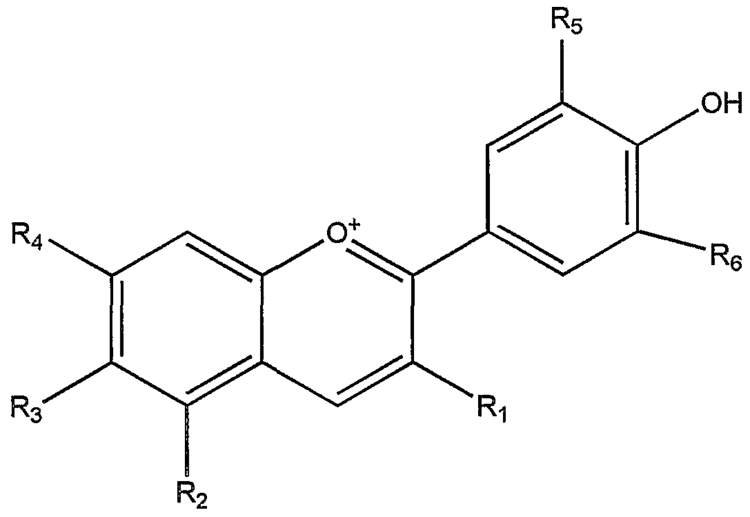
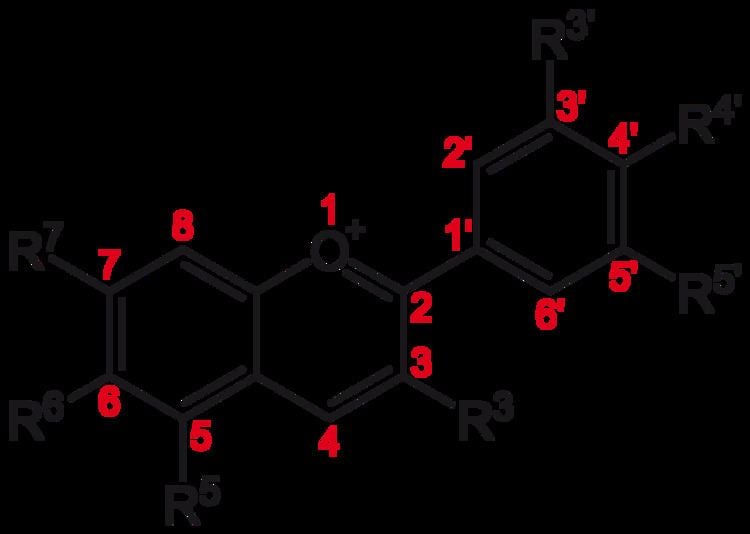
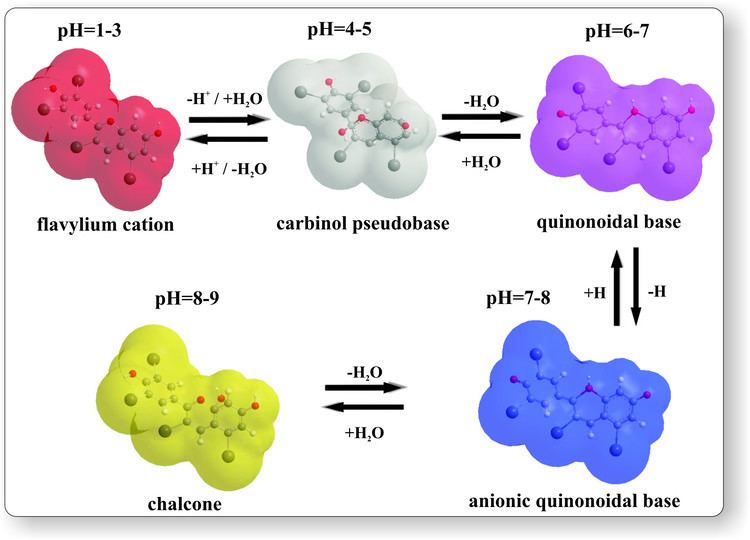
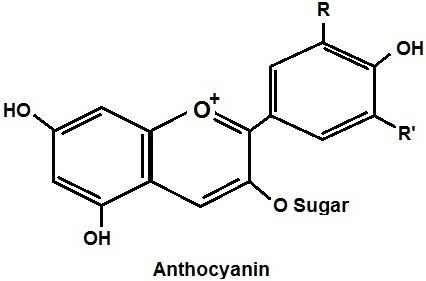
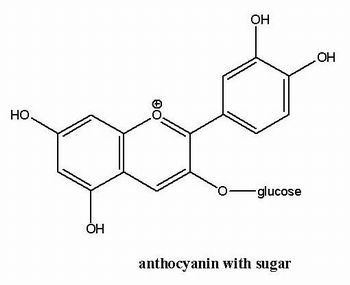
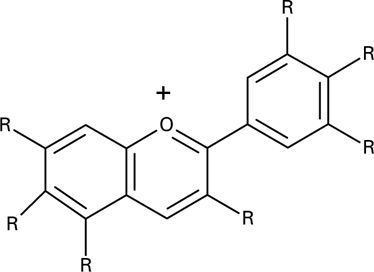
The anthocyanins, anthocyanidins with sugar group(s), are mostly 3-glucosides of the anthocyanidins. The anthocyanins are subdivided into the sugar-free anthocyanidin aglycones and the anthocyanin glycosides. As of 2003, more than 400 anthocyanins had been reported while more recent literature (early 2006), puts the number at more than 550 different anthocyanins. The difference in chemical structure that occurs in response to changes in pH is the reason why anthocyanins are often used as pH indicators, as they change from red in acids to blue in bases.
Stability
Anthocyanins are thought to be subject to physiochemical degradation in vivo and in vitro. Structure, pH, temperature, light, oxygen, metal ions, intramolecular association, and intermolecular association with other compounds (copigments, sugars, proteins, degradation products, etc.) are generally known to affect the color and stability of anthocyanins. B-ring hydroxylation status and pH have been shown to mediate the degradation of anthocyanins to their phenolic acid and aldehyde constituents. Indeed, significant portions of ingested anthocyanins are likely to degrade to phenolic acids and aldehyde in vivo, following consumption. This characteristic confounds scientific isolation of specific anthocyanin mechanisms in vivo.
pH
Anthocyanins are generally degraded at higher pH. However, some anthocyanins, such as petanin (Petunidin 3-[6-O-(4-O-(E)-p-coumaroyl-O-α-L-rhamnopyranosyl)-β-D-glucopyranoside]-5-O-β-D-glucopyranoside), are resistant to degradation at pH 8 and can be used as a food colorant.
Use as environmental pH indicator
Anthocyanins can be used as pH indicators because their color changes with pH; they are red or pink in acidic solutions (pH < 7), purple in neutral solutions (pH ~ 7), greenish-yellow in alkaline solutions (pH > 7), and colorless in very alkaline solutions, where the pigment is completely reduced.
Biosynthesis
- Anthocyanin pigments are assembled like all other flavonoids from two different streams of chemical raw materials in the cell:
- One stream involves the shikimate pathway to produce the amino acid phenylalanine. (see phenylpropanoids)
- The other stream produces three molecules of malonyl-CoA, a C3 unit from a C2 unit (acetyl-CoA).
- These streams meet and are coupled together by the enzyme chalcone synthase, which forms an intermediate chalcone-like compound via a polyketide folding mechanism that is commonly found in plants.
- The chalcone is subsequently isomerized by the enzyme chalcone isomerase to the prototype pigment naringenin.
- Naringenin is subsequently oxidized by enzymes such as flavanone hydroxylase, flavonoid 3' hydroxylase and flavonoid 3' 5'-hydroxylase.
- These oxidation products are further reduced by the enzyme dihydroflavonol 4-reductase to the corresponding colorless leucoanthocyanidins.
- Leucoanthocyanidins were once believed to be the immediate precursors of the next enzyme, a dioxygenase referred to as anthocyanidin synthase or leucoanthocyanidin dioxygenase. Flavan-3-ols, the products of leucoanthocyanidin reductase (LAR), have been recently shown to be their true substrates.
- The resulting unstable anthocyanidins are further coupled to sugar molecules by enzymes such as UDP-3-O-glucosyltransferase to yield the final relatively stable anthocyanins.
More than five enzymes are thus required to synthesize these pigments, each working in concert. Even a minor disruption in any of the mechanism of these enzymes by either genetic or environmental factors would halt anthocyanin production. While the biological burden of producing anthocyanins is relatively high, plants benefit significantly from environmental adaptation, disease tolerance, and pest tolerance provided by anthocyanins.
In anthocyanin biosynthetic pathway, L-phenylalanine is converted to naringenin by phenylalanine ammonialyase (PAL), cinnamate 4-hydroxylase (C4H), 4-coumarate CoA ligase (4CL), chalcone synthase (CHS) and chalcone isomerase (CHI). And then, the next pathway is catalyzed the formation of complex aglycone and anthocyanin composition by flavanone 3-hydroxylase (F3H), flavonoid 3'-hydroxylase (F3'H), dihydroflavonol 4-reductase (DFR), anthocyanidin synthase (ANS), UDP-glucoside: flavonoid glucosyltransferase (UFGT) and methyl transferase (MT). Among those, UFGT is divided into UF3GT and UF5GT, which are responsible for the glucosylation of anthocyanin to produce stable molecules.
In Arabidopsis thaliana, two glycosyltransferases, UGT79B1 and UGT84A2, are involved in the anthocyanin biosynthetic pathway. The UGT79B1 protein converts Cyanidin 3-O-glucoside to cyanidin 3-O-xylosyl(1→2)glucoside. UGT84A2 encodes sinapic acid: UDP-glucosyltransferase.
Genetic analysis
The phenolic metabolic pathways and enzymes can be studied by mean of transgenesis of genes. The Arabidopsis regulatory gene in the production of anthocyanin pigment 1 (AtPAP1) can be expressed in other plant species.
Dye-sensitized solar cells
Anthocyanins have been used in organic solar cells because of their ability to convert light energy into electrical energy. The many benefits to using dye-sensitized solar cells instead of traditional p-n junction silicon cells include lower purity requirements and abundance of component materials, as well as the fact they can be produced on flexible substrates, making them amenable to roll-to-roll printing processes.
Research
Anthocyanins fluoresce, enabling a tool for plant cell research to allow live cell imaging without a requirement for other fluorophores.
As of 2016, there are no substantial clinical trials to indicate that dietary anthocyanins lower the risk against human diseases.
Use as visual markers to mark genetically modified materials
Anthocyanin production can be engineered into genetically modified materials to enable their visual identification.
