3DMet B00883 Formula C3H8O Molar mass 60.09502 g/mol IUPAC ID Propan-1-ol Classification Alcohol | Appearance Colorless liquid Boiling point 97 °C Density 803 kg/m³ Melting point -126 °C | |
 | ||
Related compounds | ||
Organic conversion 2 propanol to 1 propanol secondary alcohol to primary alcohol
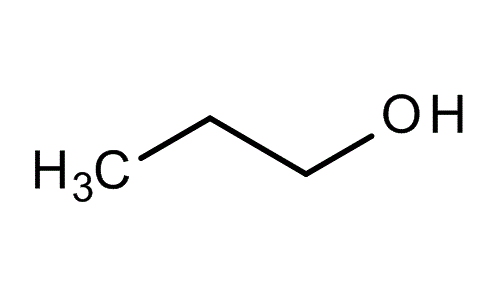
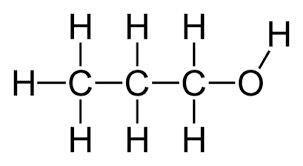
1-Propanol, is a primary alcohol with the formula CH3CH2CH2OH (sometimes represented as PrOH or n-PrOH). This colorless liquid is also known as propan-1-ol, 1-propyl alcohol, n-propyl alcohol, and n-propanol. It is an isomer of isopropanol (2-propanol, isopropyl alcohol). It is formed naturally in small amounts during many fermentation processes and used as a solvent in the pharmaceutical industry mainly for resins and cellulose esters.
Contents
- Organic conversion 2 propanol to 1 propanol secondary alcohol to primary alcohol
- Espectro de infrarrojo de alcoholes 1 propanol
- Chemical properties
- Preparation
- Safety
- Inhalation
- Propanol as fuel
- References
Espectro de infrarrojo de alcoholes 1 propanol
Chemical properties

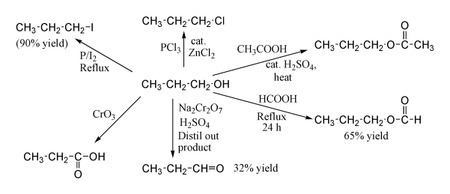
1-Propanol shows the normal reactions of a primary alcohol. Thus it can be converted to alkyl halides; for example red phosphorus and iodine produce n-propyl iodide in 80% yield, while PCl3 with catalytic ZnCl2 gives 1-chloropropane. Reaction with acetic acid in the presence of an H2SO4 catalyst under Fischer esterification conditions gives propyl acetate, while refluxing propanol overnight with formic acid alone can produce propyl formate in 65% yield. Oxidation of 1-propanol with Na2Cr2O7 and H2SO4 gives only a 36% yield of propionaldehyde, and therefore for this type of reaction higher yielding methods using PCC or the Swern oxidation are recommended. Oxidation with chromic acid yields propionic acid.
Preparation
1-Propanol is manufactured by catalytic hydrogenation of propionaldehyde. The propionaldehyde is itself produced via the oxo process, by hydroformylation of ethylene using carbon monoxide and hydrogen in the presence of a catalyst such as cobalt octacarbonyl or a rhodium complex.
H2C=CH2 + CO + H2 → CH3CH2CH=OCH3CH2CH=O + H2 → CH3CH2CH2OHA traditional laboratory preparation of 1-propanol involves treating n-propyl iodide with moist Ag2O.
1-Propanol was discovered in 1853 by Chancel, who obtained it by fractional distillation of fusel oil. Indeed, 1-propanol is a major constituent of fusel oil, a by-product formed from certain amino acids when potatoes or grains are fermented to produce ethanol. This process is no longer a significant source of 1-propanol.
Safety
1-Propanol is thought to be similar to ethanol in its effects on human body, but 2-4 times more potent. Oral LD50 in rats is 1870 mg/kg (compared to 7060 mg/kg for ethanol). It is metabolized into propionic acid. Effects include alcoholic intoxication and high anion gap metabolic acidosis. As of 2011, only one case of lethal 1-propanol poisoning was reported.
Inhalation
Although this method is rare, it does exist. Propanol might be much more convenient than ethanol for inhalation because of its potency with nebulizers.
Propanol as fuel
1-propanol has high octane numbers and it is suitable to engine fuel usage. However, production of propanol has been too expensive to be a common fuel. The research octane number (RON) of propanol is 118 and anti-knock index (AKI) is 108.
