Formula C3H9SnCl Melting point 38.5 °C | Molar mass 199.27 g/mol Boiling point 148 °C | |
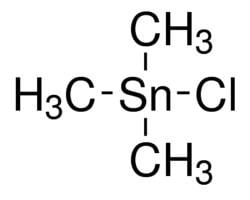 | ||
Trimethyltin chloride is an organotin compound with the formula (CH3)3SnCl. It is a white solid that is highly toxic and has a stench. It is susceptible to hydrolysis.
Contents
Synthesis
Trimethyltin chloride can be prepared by the redistribution reaction of tetramethyltin with tin tetrachloride.
SnCl4 + 3 SnMe4 → 4 Me3SnClThis is the Kocheshkov redistribution reaction. It is performed under an inert atmosphere, such as argon, typically with no solvent.
A second route to Me3SnCl involves treating the corresponding hydroxide or oxide with a halogenating agent such as hydrogen chloride or thionyl chloride (SOCl2):
Me3SnOH + HCl → Me3SnCl + H2OUses
Trimethyltin chloride is used as a source of the trimethylstannyl group. For example, it is a precursor to vinyltrimethylstannane:
CH2=CHMgBr + Me3SnCl → Me3SnCH=CH2 + MgBrClAn example of an organolithium reagent reacting with Me3SnCl to form a tin-carbon bond is:
LiCH(SiMe3)(GeMe3) + Me3SnCl → Me3SnCH(SiMe3)(GeMe3) + LiClOrganotin compounds derived from Me3SnCl are useful in organic synthesis, especially in radical chain reactions. Me3SnCl is a precursor to compounds used in PVC stabilization. Reduction of trimethyltin chloride gives tin-tin bonds.
Me3SnM + Me3SnCl → Sn2Me6 + MCl (M = metal)