AHFS/Drugs.com Monograph CAS ID 28210-41-5 | MedlinePlus a682108 | |
 | ||
Trade names Pregnancycategory US: C (Risk not ruled out) | ||
Polystyrene sulfonates are polymers derived from polystyrene but containing sulfonate functional groups. They are widely used as ion-exchange resins to remove ions such as potassium, calcium and sodium from solutions in technical or medical applications.
Contents
- Medical uses
- Contraindications
- Side effects
- Interactions
- Hyperkalemia
- Clostridium difficile associated diarrhea CDAD
- Production and chemical structure
- Alternative sulfonation methods
- Termination of tolevamer development
- Chemical uses
- Water softening
- Other uses
- References
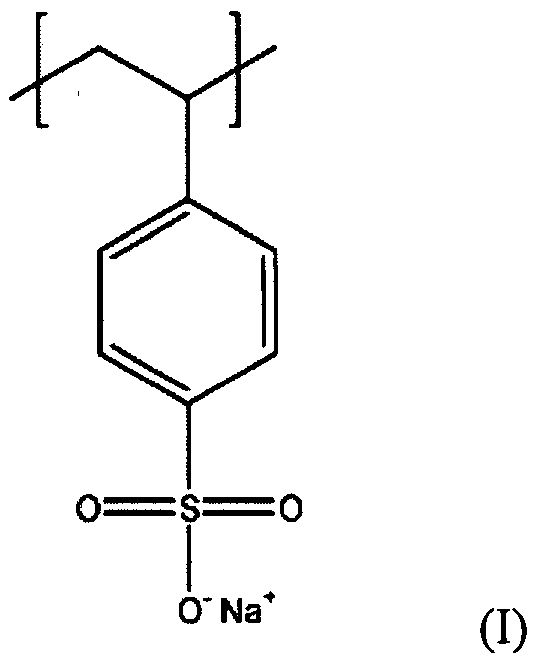
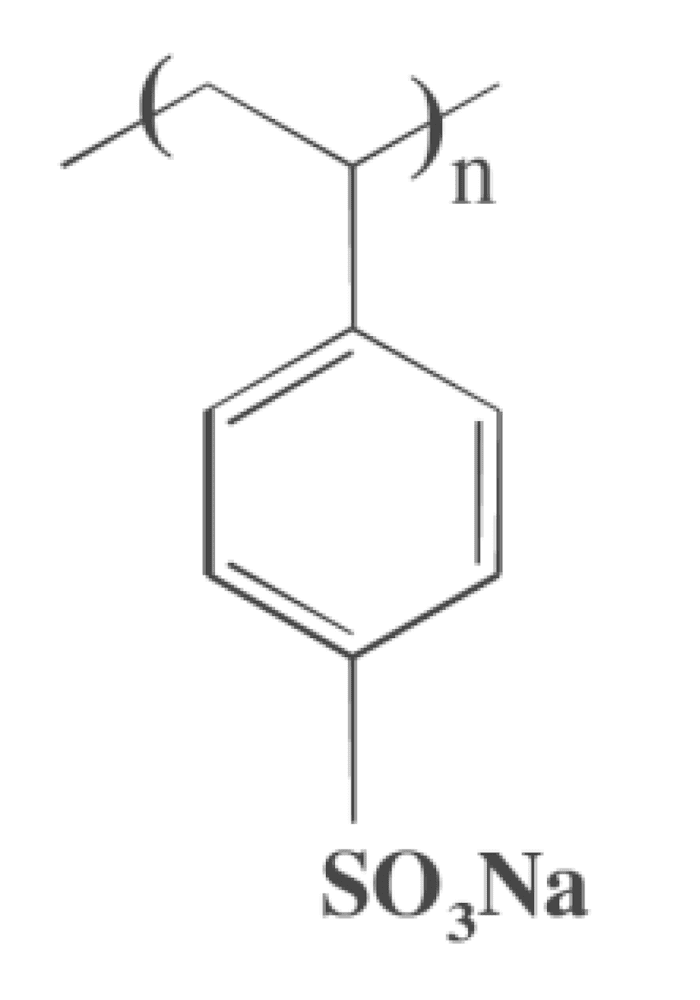
Linear ionic polymers are generally water-soluble, whereas cross-linked materials (called resins) do not dissolve in water. These polymers are classified as polysalts and ionomers.

Medical uses
polystyrene sulfonate is usually supplied in the sodium and calcium form. It is used as a potassium binder in acute and chronic kidney disease for people with hyperkalemia (abnormal high blood serum potassium levels). It however is unclear if it works and there is concern about possible side effects when it is mixed with sorbitol.
Polystyrene sulfonates are given by mouth (with a meal) or rectally, by retention enema.

Under the name tolevamer, a polystyrene sulfonate was investigated by Genzyme as a toxin binding agent for the treatment of Clostridium difficile associated diarrhoea (CDAD), but never marketed.
Contraindications
The drug is contraindicated in patients with obstructive bowel disease and in newborn children with reduced gut motility.
Side effects

Intestinal disturbances are common, including loss of appetite, nausea, vomiting, and constipation. In rare cases, it has been associated with colonic necrosis. Changes in electrolyte blood levels may occur such as hypermagnesemia, hypercalcemia, and hypokalemia.
Interactions

Polystyrene sulfonates can bind to various drugs within the digestive track and thus lower their absorption and effectiveness. Examples include lithium and thyroxine. digitalis is more likely to cause side effects in the presence of hypokalemia, which can be caused by polystyrene sulfonate. Cases of intestinal necrosis have been reported when this polymer was combined with sorbitol.
Hyperkalemia
Polystyrene sulfonates release sodium or calcium ions in the stomach in exchange for hydrogen ions, when the resin reaches the large intestine the hydrogen ions are exchanged for free potassium ions; the resin is then eliminated in the feces. The net effect is lowering the amount of potassium available for absorption into the blood and increasing the amount that is excreted via the feces. The effect is a reduction of potassium levels in the body.
Clostridium difficile associated diarrhea (CDAD)

Tolevamer was designed to bind the enterotoxins rather than attack Clostridium difficile directly. Since it has no antibiotic properties, it does not harm the gut flora. Early studies used the sodium salt, but it was soon replaced with the potassium sodium salt to prevent hypokalaemia which is often associated with diarrhoea.
Production and chemical structure
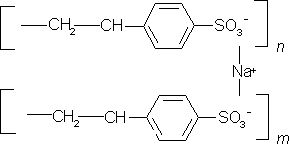
Polystyrene sulfonic acid has the idealized formula (CH2CHC6H4SO3H)n. The material is prepared by sulfonation of polystyrene:
(CH2CHC6H5)n + n SO3 → (CH2CHC6H4SO3H)nSeveral methods exist for this conversion, which can lead to varying degree of sulfonation. Usually the polystyrene is crosslinked, which keeps the polymer from dissolving. Since the sulfonic acid group (SO3H) is strongly acidic, this polymer neutralizes bases. In this way, various salts of the polymer can be prepared, leading to sodium, calcium, and other salts:
(CH2CHC6H4SO3H)n + n NaOH → (CH2CHC6H4SO3Na)n + n H2OThese ion-containing polymers are called ionomers.
Alternative sulfonation methods
Double substitutions of the phenyl rings are known to occur, even with conversions well below 100%. Crosslinking reactions are also found, where condensation of two sulfonic acid groups yields a sulfonyl crosslink. On the other hand, the use of milder conditions such as acetyl sulfate leads to incomplete sulfonation. Recently, the atom transfer radical polymerization (ATRP) of protected styrenesulfonates has been reported, leading to well defined linear polymers, as well as more complicated molecular architectures.
Termination of tolevamer development
In early 2008, a noninferiority study versus vancomycin or metronidazole for Clostridium difficile associated diarrhea (CDAD) found that about half of the patients in the tolevamer group did not complete the treatment, versus 25% in the vancomycin and 29% in the metronidazole groups.
CDAD recurrence in patients reaching clinical success was reduced significantly by tolevamer (6% recurrence rate), vancomycin (18%) and metronidazole (19%). However, the good result of tolevamer is partly due to the high drop-out rate in this group.
Since tolevamer did not reach its primary endpoint in this study, its development was halted.
Chemical uses
Polystyrene sulfonates are useful because of their ion exchange properties.
Water softening
Water softening is achieved by percolating hard water through a bed of the sodium form of cross-linked polystyrene sulfonate. The hard ions such as calcium (Ca2+) and magnesium (Mg2+) adhere to the sulfonate groups, displacing sodium ions. The resulting solution of sodium ions is softened.
Other uses
Sodium polystyrene sulfonate is used as a superplastifier in cement, as a dye improving agent for cotton, and as proton exchange membranes in fuel cell applications. In its acid form, the resin is used as a solid acid catalyst in organic synthesis.
