ICD-9-CM 747.0 DiseasesDB 9706 | ICD-10 Q25.0 OMIM 607411 MedlinePlus 001560 | |
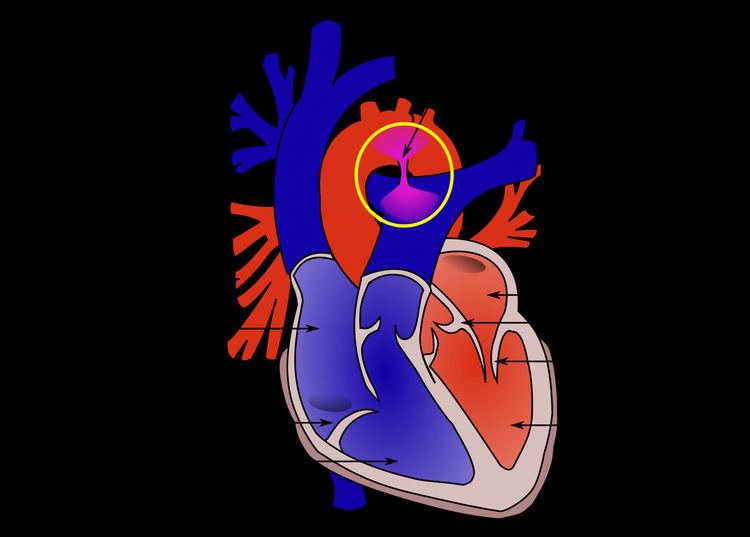 | ||
Patent ductus arteriosus (PDA) is a condition wherein the ductus arteriosus fails to close after birth.
Contents
Early symptoms are uncommon, but in the first year of life include increased 'work of breathing' and poor weight gain. An uncorrected PDA may lead to congestive heart failure with increasing age.
The ductus arteriosus is a fetal blood vessel that closes soon after birth. In a PDA, the vessel does not close and remains "patent" (open), resulting in irregular transmission of blood between the aorta and the pulmonary artery. PDA is common in newborns with persistent respiratory problems such as hypoxia, and has a high occurrence in premature newborns. Premature newborns are more likely to be hypoxic and have PDA due to underdevelopment of the heart and lungs.
A PDA allows a portion of the oxygenated blood from the left heart to flow back to the lungs by flowing from the aorta (which has higher pressure) to the pulmonary artery. If this shunt is substantial, the neonate becomes short of breath: the additional fluid returning to the lungs increases lung pressure, which in turn increases the energy required to inflate the lungs. This uses more calories than normal and often interferes with feeding in infancy. This condition, as a constellation of findings, is called congestive heart failure.
In some congenital heart defects (such as in transposition of the great vessels) a PDA may need to remain open, as it is the only way that oxygenated blood can mix with deoxygenated blood. In these cases, prostaglandins are used to keep the DA open until surgical correction of the heart defect is completed.
Signs and symptoms
Common symptoms include:
Patients typically present in good health, with normal respirations and heart rate. If the PDA is moderate or large, widened pulse pressure and bounding peripheral pulses are frequently present, reflecting increased left ventricular stroke volume and diastolic run-off of blood into the (initially lower-resistance) pulmonary vascular bed. Prominent suprasternal and carotid pulsations may be noted secondary to increased left ventricular stroke volume.
Cause
A PDA is sometimes idiopathic. Known risk factors include:
Diagnosis
PDA is usually diagnosed using noninvasive techniques. Echocardiography (in which sound waves are used to capture the motion of the heart) and associated Doppler studies are the primary methods of detecting PDA. Electrocardiography (ECG), in which electrodes are used to record the electrical activity of the heart, is not particularly helpful as no specific rhythms or ECG patterns can be used to detect PDA.
A chest X-ray may be taken, which reveals overall heart size (as a reflection of the combined mass of the cardiac chambers) and the appearance of blood flow to the lungs. A small PDA most often accompanies a normal-sized heart and normal blood flow to the lungs. A large PDA generally accompanies an enlarged cardiac silhouette and increased blood flow to the lungs.
Normal anatomy
In the developing fetus, the DA is the vascular connection between the pulmonary artery and the aortic arch that allows most of the blood from the right ventricle to bypass the fetus' lungs, which are fluid-filled and compressed. During fetal development, this shunt protects the right ventricle from pumping against the high resistance in the lungs, which can lead to right ventricular failure if the DA closes in utero.
When the newborn takes his or her first breath, the lungs open and pulmonary vascular resistance decreases. After birth, the lungs release bradykinin to constrict the smooth muscle wall of the DA and reduce blood flow as it narrows and then completely closes. In most newborns with a patent ductus arteriosus, the blood flow is reversed from that of in utero flow; i.e., the blood flow is from the higher-pressure aorta to the now lower-pressure pulmonary arteries.
In normal newborns, the DA is substantially narrowed within 12–24 hours after birth, and seals completely after three weeks.[Needs Citation] The primary stimulus for closure of the DA is an increase in neonatal blood oxygen content. Withdrawal from maternal circulating prostaglandins also contributes to ductal closure. The residual scar tissue from the fibrotic remnants of the DA, called the ligamentum arteriosum, remains in the normal adult heart.
Prevention
Some evidence suggests that indomethacin administration on the first day of life to all preterm infants reduces the risk of developing a PDA and the complications associated with PDA. Indomethacin treatment in premature infants also may reduce the need for surgical intervention.
Treatment
Neonates without adverse symptoms may simply be monitored as outpatients, while symptomatic PDA can be treated with both surgical and non-surgical methods. Surgically, the DA may be closed by ligation (though support in premature infants is mixed), either manually tied shut, or with intravascular coils or plugs that leads to formation of a thrombus in the DA.
Devices developed by Franz Freudenthal block the blood vessel with woven structures of nitinol wire.
Because prostaglandin E2 is responsible for keeping the DA open, NSAIDS (which can inhibit prostaglandin synthesis) such as indomethacin or a special form of ibuprofen have been used to initiate PDA closure. Recent findings from a systematic review concluded that, for closure of a PDA in preterm and/or low birth weight infants, ibuprofen is as effective as Indomethacin. It also causes fewer side effects (such as transient renal insufficiency) and reduces the risk of necrotising enterocolitis. Another recent review showed that paracetamol may be effective for closure of a PDA in preterm infants.
More recently, PDAs can be closed by percutaneous interventional method (avoiding open heart surgery). A platinum coil can be deployed via a catheter through the femoral vein or femoral artery, which induces thrombosis (coil embolization). Alternatively, a PDA occluder device, composed of nitinol mesh, is deployed from the pulmonary artery through the PDA.
Prognosis
If left untreated, the disease may progress from left-to-right shunt (acyanotic heart) to right-to-left shunt (cyanotic heart), called Eisenmenger's syndrome. Pulmonary hypertension is a potential long-term outcome, which may require a heart and/or lung transplant. Another complication of PDA is intraventricular hemorrhage.
History
Robert E. Gross, MD performed the first successful ligation of a patent ductus arteriosus on an eight-year-old girl at Children's Hospital Boston in 1938.
Adult
Since PDA is usually identified in infants, it is less common in adults, but it can have serious consequences, and is usually corrected surgically upon diagnosis.
