Trade names Rogaine Pregnancycategory C Formula C9H15N5O Melting point 248 °C Soluble in Water | AHFS/Drugs.com Monograph CAS ID 38304-91-5 Molar mass 209.251 g/mol | |
 | ||
ATC code C02DC01 (WHO) D11AX01 (WHO) Legal status UK: P (Pharmacy medicines) (for topical use, otherwise POM. Cannot be prescribed on the NHS) IUPAC ID 6-piperidin-1-ylpyrimidine-2,4-diamine 3-oxide | ||
Minoxidil is an antihypertensive vasodilator medication. It also slows hair loss and promotes hair regrowth in some people. It is available as a generic medication and over the counter for the treatment of androgenic alopecia, a form of hair loss, in men and women.
Contents
- Minoxidil with hair loss treatments
- Medical uses
- Side effects
- Mechanism of action
- Initial application
- Hair growth
- Trade names
- Research
- References

Minoxidil with hair loss treatments
Medical uses
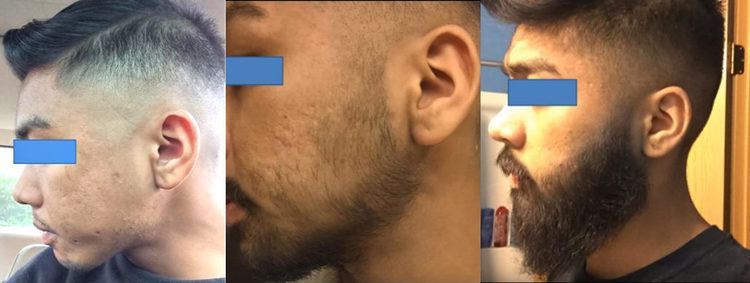
Minoxidil, applied topically, is widely used for the treatment of hair loss. It is effective in helping promote hair growth in both men and women with androgenic alopecia. About 40% of men experience hair regrowth after 3–6 months. Minoxidil must be used indefinitely for continued support of existing hair follicles and the maintenance of any experienced hair regrowth.
Its effect in people with alopecia areata is unclear.
Side effects

Minoxidil is generally well tolerated, but common side effects include burning or irritation of the eye, itching, redness or irritation at the treated area, as well as unwanted hair growth elsewhere on the body. Exacerbation of hair loss/alopecia has been reported. Severe allergic reactions may include rash, hives, itching, difficulty breathing, tightness in the chest, swelling of the mouth, face, lips, or tongue, chest pain, dizziness, fainting, tachycardia, headache, sudden and unexplained weight gain, or swelling of the hands and feet. Temporary hair loss is a common side effect of minoxidil treatment. Manufacturers note that minoxidil-induced hair loss is a common side effect and describe the process as "shedding".
Alcohol and propylene glycol present in some topical preparations may dry the scalp, resulting in dandruff and contact dermatitis.
Side effects of oral minoxidil may include swelling of the face and extremities, rapid and irregular heartbeat, lightheadedness, cardiac lesions, and focal necrosis of the papillary muscle and subendocardial areas of the left ventricle. There have been cases of allergic reactions to minoxidil or the non-active ingredient propylene glycol, which is found in some topical minoxidil formulations. Pseudoacromegaly is an extremely rare side effect reported with large doses of oral minoxidil.
Mechanism of action

The mechanism by which minoxidil promotes hair growth is not fully understood. Minoxidil is a potassium channel opener, causing hyperpolarization of cell membranes. Hypothetically, by widening blood vessels and opening potassium channels, it allows more oxygen, blood, and nutrients to the follicles. This may cause follicles in the telogen phase to shed, which are then replaced by thicker hairs in a new anagen phase. Minoxidil is a prodrug that is converted by sulfation via the sulfotransferase enzyme SULT1A1 to its active form, minoxidil sulfate. Several studies demonstrated that the activity of sulfotransferase in hair follicles predict minoxidil response in the treatment of hair loss.

Minoxidil is less effective when there is a large area of hair loss. In addition, its effectiveness has largely been demonstrated in younger men who have experienced hair loss for less than 5 years. Minoxidil use is indicated for central (vertex) hair loss only. Minoxidil is also a vasodilator. Two clinical studies are being conducted in the US for a medical device that may allow patients to determine if they are likely to fail minoxidil therapy.
Initial application
Minoxidil was developed in the late 1950s by the Upjohn Company (later became part of Pfizer) to treat ulcers. In trials using dogs, the compound did not cure ulcers, but proved to be a powerful vasodilator. Upjohn synthesized over 200 variations of the compound, including the one it developed in 1963 and named minoxidil. These studies resulted in FDA approving minoxidil (with the trade name 'Loniten') in the form of oral tablets to treat high blood pressure in 1979.
Hair growth
When Upjohn received permission from the FDA to test the new drug as medicine for hypertension they approached Charles A. Chidsey MD, Associate Professor of Medicine at the University of Colorado School of Medicine., who conducted two studies, the second study showing unexpected hair growth. Puzzled by this side-effect, Chidsey consulted Guinter Kahn and discussed the possibility of using minoxidil for treating hair loss.
Kahn along with his colleague Paul J. Grant MD obtained a certain amount of the drug and conducted their own research, apparently without notifying Upjohn or Chidsey. The two doctors had been experimenting with a 1% solution of minoxidil mixed with several alcohol-based liquids. They tried to patent the drug for hair loss prevention, but found that Upjohn had already done this. A decade-long trial between Kahn and Upjohn ended with Kahn's name included in a consolidated patent (U.S. #4,596,812 Charles A Chidsey, III and Guinter Kahn) in 1986 and royalties from the company to both Kahn and Grant.
Meantime the effect of minoxidil on hair loss prevention was so clear that in the 80's physicians were prescribing Loniten off-label to their balding patients.
In August 18, 1988 the FDA finally approved the drug for treating baldness in men under the trade name 'Rogaine' (FDA rejected Upjohn's first choice, Regain, as misleading). The agency concluded that although "the product will not work for everyone", 39 percent of the men studied had "moderate to dense hair growth on the crown of the head".
In 1991 Upjohn made the product available for women.
On February 12, 1996 FDA approved both the over-the-counter sale of the drug and the production of generic formulations of minoxidil. Upjohn replied to that by lowering prices to half the price of the prescription drug and by releasing a prescription 5% formula of Rogaine in 1997.
In 1998 a 5% formulation of minoxidil was approved for nonprescription sale by the FDA.
As of 2014 it was the only topical product that is FDA-approved for androgenic hair loss.
The drug is available over the counter in the United Kingdom.
Trade names
Minoxidil is marketed under many trade names worldwide.
Research
Alternate formulations of minoxidil have been explored in order to reduce contact dermatitis from the alcohol and propylene glycol vehicle.
