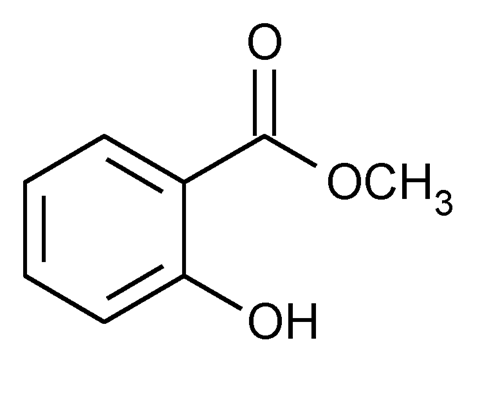
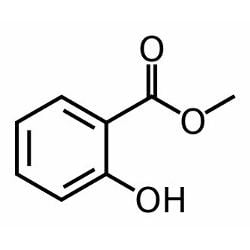
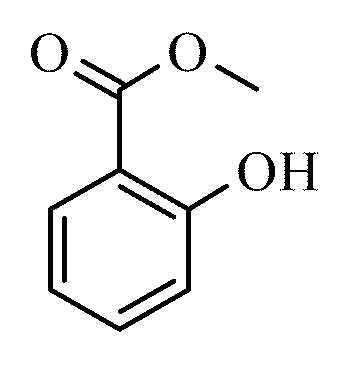
Formula C8H8O3 Boiling point 220 °C Melting point -9 °C | Molar mass 152.1494 g/mol Density 1.17 g/cm³ IUPAC ID Methyl 2-hydroxybenzoate | |
 | ||
Methyl salicylate (oil of wintergreen or wintergreen oil) is an organic ester naturally produced by many species of plants, particularly wintergreens. It is also synthetically produced, used as a fragrance, in foods and beverages, and in liniments.
Contents
The compound methyl salicylate was first isolated (from the plant Gaultheria procumbens) in 1843 by the French chemist Auguste André Thomas Cahours (1813–1891), who identified it as an ester of salicylic acid and methanol.
Natural occurrence

This compound is probably produced as an anti-herbivore defense. If the plant is infected with herbivorous insects, the release of methyl salicylate may function as an aid in the recruitment of beneficial insects to kill the herbivorous insects. Aside from its toxicity, methyl salicylate may also be used by plants as a pheromone to warn other plants of pathogens such as tobacco mosaic virus.
Numerous plants produce methyl salicylate in very small amounts. Some plants, such as the following, produce more:

Commercial production
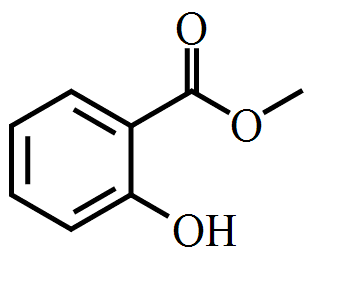
Methyl salicylate can be produced by esterifying salicylic acid with methanol. Commercial methyl salicylate is now synthesized, but in the past, it was commonly distilled from the twigs of Betula lenta (sweet birch) and Gaultheria procumbens (eastern teaberry or wintergreen).
Uses
Safety and toxicity
In pure form, methyl salicylate is harmful, when taken orally. A single teaspoon (5 ml) of methyl salicylate contains approximately 6 g of salicylate, which is equivalent to almost twenty 300 mg aspirin tablets (5 mL * 1.174 g/mL = 5.87 g). The lowest published lethal dose is 101 mg/kg body weight in adult humans, (or 7.07 grams for a 70-kg adult). It has proven fatal to small children in doses as small as 4 ml. A seventeen-year-old cross-country runner at Notre Dame Academy on Staten Island died in April 2007 after her body absorbed methyl salicylate through excessive use of topical muscle-pain relief products.
Most instances of human toxicity due to methyl salicylate are a result of over-application of topical analgesics, especially involving children. Some people have intentionally ingested large amounts of oil of wintergreen. Salicylate, the major metabolite of methyl salicylate, may be quantitated in blood, plasma or serum to confirm a diagnosis of poisoning in hospitalized patients or to assist in an autopsy.
