Appearance Colorless gas Formula C4H10 IUPAC ID isobutane Molar mass 58.12 g/mol Classification Alkane | Related alkane Boiling point -11.7 °C Density 2.51 kg/m³ Melting point -159.6 °C | |
 | ||
Thermodynamicdata Phase behavioursolid–liquid–gas | ||
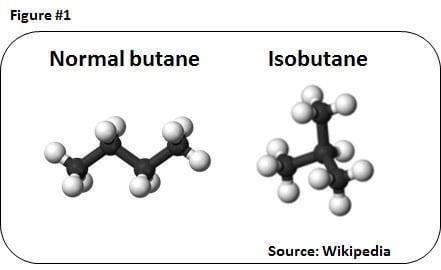
Isobutane vs butane chemistry lessons
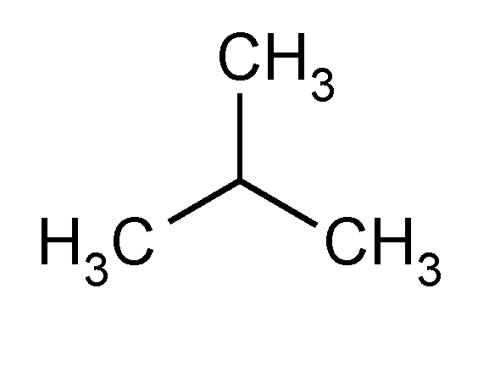
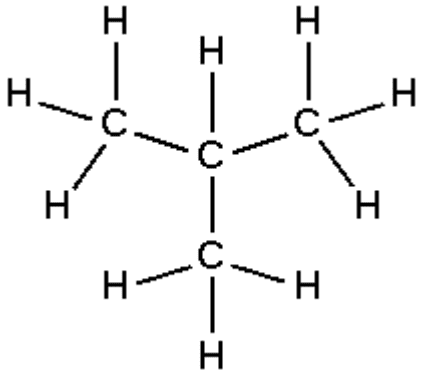
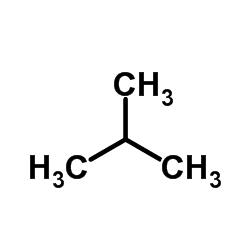
Isobutane (i-butane), also known as methylpropane, is a chemical compound with molecular formula C
4H
10 and is an isomer of butane. It is the simplest alkane with a tertiary carbon. Concerns with depletion of the ozone layer by freon gases have led to increased use of isobutane as a gas for refrigeration systems, especially in domestic refrigerators and freezers, and as a propellant in aerosol sprays.
Contents
- Isobutane vs butane chemistry lessons
- How to identify the charge of an ion chemistry lessons
- Nomenclature
- Uses
- Refrigerant use
- References

When used as a refrigerant or a propellant, isobutane is also known as R-600a. Some portable camp stoves use a mixture of isobutane with propane, usually 80:20. Isobutane is used as a feedstock in the petrochemical industry, for example in the synthesis of isooctane.

How to identify the charge of an ion chemistry lessons
Nomenclature

The traditional name isobutane was still retained in the 1993 IUPAC recommendations, but is no longer recommended according to the 2013 recommendations. Since the longest continuous chain in isobutane contains only three carbon atoms, the preferred IUPAC name is 2-methylpropane but the locant (2-) is typically omitted in general nomenclature as redundant; C2 is the only position on a propane chain where a methyl substituent can be located without altering the main chain and forming the constitutional isomer n-butane.
Uses
Isobutane is used as a refrigerant. The use in refrigerators started in 1993 when Greenpeace presented the Greenfreeze project with the German company Foron. In this regard, blends of pure, dry "isobutane" (R-600a) (that is, isobutane mixtures) have negligible ozone depletion potential and very low global warming potential (having a value of 3.3 times the GWP of carbon dioxide) and can serve as a functional replacement for R-12, R-22, R-134a, and other chlorofluorocarbon or hydrofluorocarbon refrigerants in conventional stationary refrigeration and air conditioning systems.
In the Chevron Phillips slurry process for making high-density polyethylene, isobutane is used as a diluent. As the slurried polyethylene is removed, isobutane is "flashed" off, and condensed, and recycled back into the loop reactor for this purpose.
Isobutane is also used as a propellant for aerosol cans and foam products.
Isobutane is used as part of blended fuels, especially common in fuel canisters used for camping.
Isobutane is also used as a raw material in the manufacture of propylene oxide, where it is oxidized to tert-butyl hydroperoxide and subsequently reacted with propylene to yield propylene oxide. The tert-butanol that results as a by-product is typically used to make gasoline additives such as methyl tert-butyl ether (MTBE).
Refrigerant use
As a refrigerant, isobutane has an explosion risk in addition to the hazards associated with non-flammable CFC refrigerants. Reports surfaced in late 2009 suggesting the use of isobutane as a refrigerant in domestic refrigerators was potentially dangerous. One refrigerator explosion reported in the United Kingdom was suspected to have been caused as a result of isobutane leaking into the refrigerator cabinet and being ignited by sparks in the electrical system. Although unclear how serious this could be, at the time this report came out it was estimated 300 million refrigerators worldwide use isobutane as a refrigerant.
Substitution of this refrigerant for motor vehicle air conditioning systems not originally designed for isobutane is widely prohibited or discouraged, on the grounds that using flammable hydrocarbons in systems originally designed to carry non-flammable refrigerant presents a significant risk of fire or explosion.
Vendors and advocates of hydrocarbon refrigerants argue against such bans on the grounds that there have been very few such incidents relative to the number of vehicle air conditioning systems filled with hydrocarbons.
