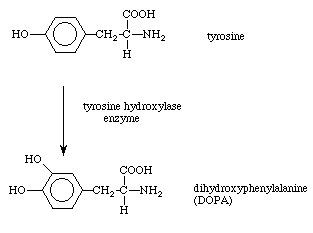
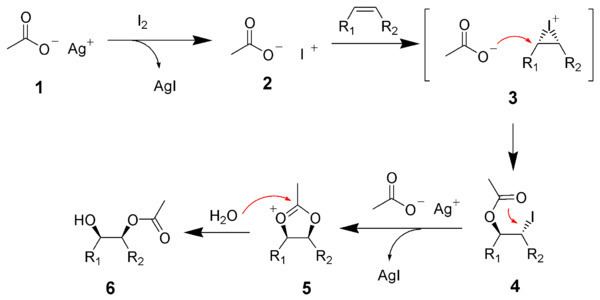
 | ||
Hydroxylation of an aliphatic compound
Hydroxylation is a chemical process that introduces a hydroxyl group (-OH) into an organic compound. In biochemistry, hydroxylation reactions are often facilitated by enzymes called hydroxylases. Hydroxylation is the first step in the oxidative degradation of organic compounds in air. It is extremely important in detoxification since hydroxylation converts lipophilic compounds into water-soluble (hydrophilic) products that are more readily removed by the kidneys or liver and excreted. Some drugs (for example, steroids) are activated or deactivated by hydroxylation.
Contents
- Hydroxylation of an aliphatic compound
- Chemical concepts
- Biological hydroxylation
- Hydroxylation of proteins
- Examples of hydroxylases
- References

Chemical concepts
The hydroxylation process involves conversion of a CH group into a COH group. Hydroxylation is an oxidative process. The oxygen that is inserted into the C-H bond is usually derived from atmospheric oxygen (O2). Since O2 itself is a slow hydroxylating agent, catalysts are required to accelerate the pace of the process.
Biological hydroxylation
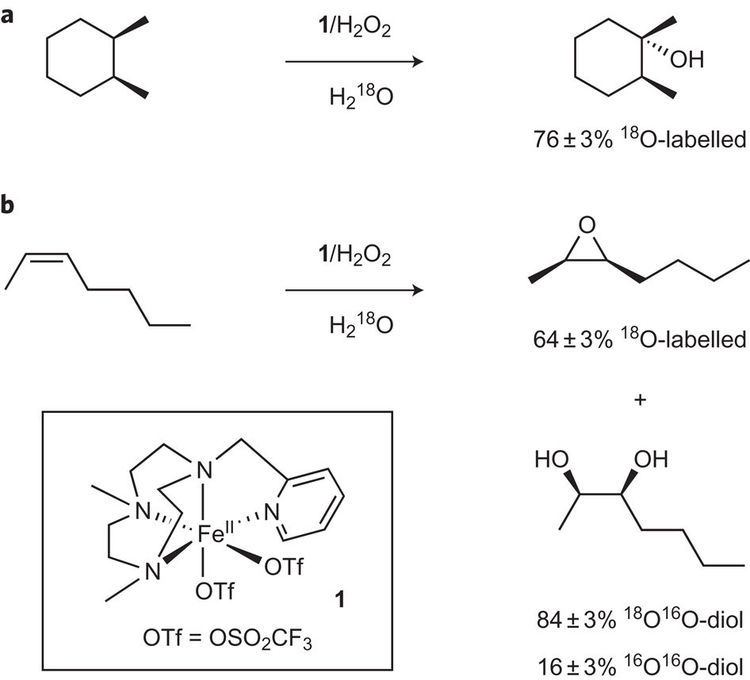
The principal hydroxylation agent in nature is cytochrome P-450, hundreds of variations of which are known. Other hydroxylating agents include flavins.
Hydroxylation of proteins
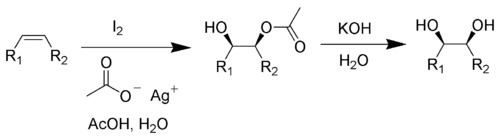
The most frequently hydroxylated residue in human proteins is proline. This is due to the fact that collagen makes up about 25-35% of the protein in our bodies and contains a hydroxyproline at almost every 3rd residue in its amino acid sequence. Hydroxylation occurs at the γ-C atom, forming hydroxyproline (Hyp), which stabilizes the secondary structure of collagen due to the strong electronegative effects of oxygen. Proline hydroxylation is also a vital component of hypoxia response via hypoxia inducible factors. In some cases, proline may be hydroxylated instead on its β-C atom. Lysine may also be hydroxylated on its δ-C atom, forming hydroxylysine (Hyl).
These three reactions are catalyzed by very large, multi-subunit enzymes prolyl 4-hydroxylase, prolyl 3-hydroxylase and lysyl 5-hydroxylase, respectively. These reactions require iron (as well as molecular oxygen and α-ketoglutarate) to carry out the oxidation, and use ascorbic acid (vitamin C) to return the iron to its oxidized state. Deprivation of ascorbate leads to deficiencies in proline hydroxylation, which leads to less stable collagen, which can manifest itself as the disease scurvy. Since citrus fruits are rich in vitamin C, British sailors were given limes to combat scurvy on long ocean voyages; hence, they were called "limeys".
Examples of hydroxylases