Formula C9H9NO3 Boiling point 240 °C | Molar mass 179.17 g/mol Density 1.37 g/cm³ | |
 | ||
Hippuric acid meaning
Hippuric acid (Gr. hippos, horse, ouron, urine) is a carboxylic acid found in the urine of horses and other herbivores. Hippuric acid crystallizes in rhombic prisms which are readily soluble in hot water, melt at 187 °C, and decompose at about 240 °C. High concentrations of hippuric acid may also indicate a toluene intoxication; however, scientists have called this correlation into question because there are other variables that influence levels of hippuric acid. When many aromatic compounds such as benzoic acid and toluene are taken internally, they are converted to hippuric acid by reaction with the amino acid, glycine.
Contents
10 hippuric acid or benzoyl glycin
Synthesis
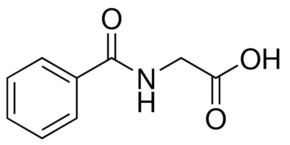
A modern synthesis of hippuric acid involves the acylation of glycine with benzoyl chloride:
Reactions
Hippuric acid is readily hydrolysed by hot caustic alkalis to benzoic acid and glycine. Nitrous acid converts it into benzoyl glycolic acid, C6H5C(=O)OCH2CO2H. Its ethyl ester reacts with hydrazine to form hippuryl hydrazine, C6H5CONHCH2CONHNH2, which was used by Theodor Curtius for the preparation of hydrazoic acid.
History
Justus von Liebig showed in 1829 that hippuric acid differed from benzoic acid, and in 1834 determined its constitution, while in 1853 French chemist Victor Dessaignes (1800–1885) synthesized it by the action of benzoyl chloride on the zinc salt of glycine. It was also formed by heating benzoic anhydride with glycine, and by heating benzamide with monochloroacetic acid.
