Density 1.75 g/cm³ Molar mass 116.52 g/mol Melting point -80 °C | Formula HSO3Cl Boiling point 151 °C Classification Sulfonic acid | |
 | ||
Related compounds Appearance colorless liquid that fumes in air | ||
Chlorosulfuric acid (IUPAC name: sulfurochloridic acid) is the inorganic compound with the formula HSO3Cl. It is also known as chlorosulfonic acid, being the sulfonic acid of chlorine. It is a distillable, colorless liquid which is hygroscopic and a powerful lachrymator.
Contents
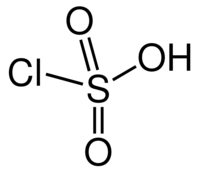
Structure and properties
Chlorosulfuric acid is a tetrahedral molecule. The formula is more descriptively written SO2(OH)Cl, but HSO3Cl is traditional. It is an intermediate, chemically and conceptually, between sulfuryl chloride (SO2Cl2) and sulfuric acid (H2SO4). The compound is rarely obtained pure. Upon standing with excess sulfur trioxide, it decomposes to pyrosulfuryl chlorides:
2 ClSO3H + SO3 → H2SO4 + S2O5Cl2Synthesis
The industrial synthesis entails the reaction of hydrogen chloride with a solution of sulfur trioxide in sulfuric acid:
It can also be prepared by chlorination of sulfuric acid, written here for pedagogical purposes as SO2(OH)2, vs. the usual format H2SO4:
The latter method is more suited for laboratory-scale operations.
Applications
ClSO2OH is used to prepare alkyl sulfates, which are useful as detergents and as chemical intermediates:
An early synthesis of saccharin begins with the reaction of toluene with ClSO2OH to give the ortho- and para-toluenesulfonyl chloride derivatives:
Oxidation of the ortho isomer gives the benzoic acid derivative that then is cyclized with ammonia and neutralized with base to afford saccharin.
Chlorosulphonic acid has been used as an anti-contrail agent in Ryan Model 147 reconnaissance drones, and to produce smoke screens.
Safety
ClSO3H reacts violently with water to yield sulfuric acid and HCl, commonly seen as vapors fuming from the liquid. Precautions, such as proper ventilation, associated with HCl should be observed.
