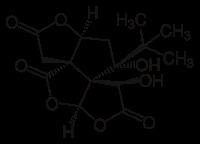
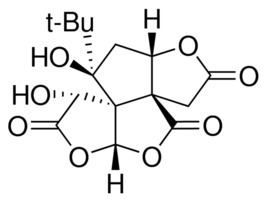
Routes ofadministration oral CAS Number 33570-04-6 IUPHAR/BPS 2366 Molar mass 326.299 g/mol | Legal status In general: legal PubChem CID 73581 ChemSpider 21106418 | |
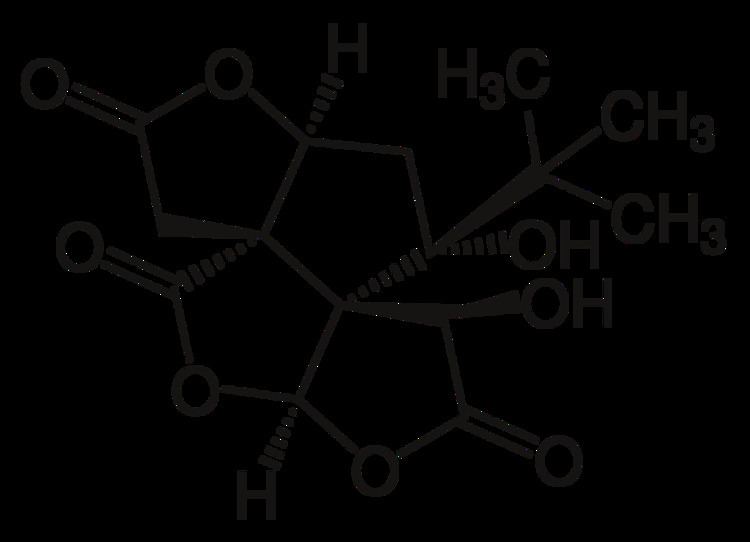 | ||
Camag hptlc application tutorial quantification of ginkgolides and bilobalide by hptlc
Bilobalide is a biologically active terpenic trilactone present in Ginkgo biloba.
Contents
- Camag hptlc application tutorial quantification of ginkgolides and bilobalide by hptlc
- How to pronounce bilobalide
- Chemistry
- Biosynthesis
- Pharmacology
- References
How to pronounce bilobalide
Chemistry
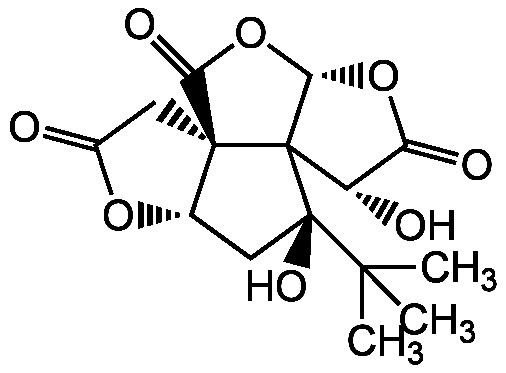
Bilobalide is a main constituent of the terpenoids found in Ginkgo leaves. It also exists in minor amounts in the roots. It is a sesquiterpenoid, i.e. it has a 15-carbon skeleton. Its exact synthesis pathway from farnesyl pyrophosphate is still unknown.
Biosynthesis
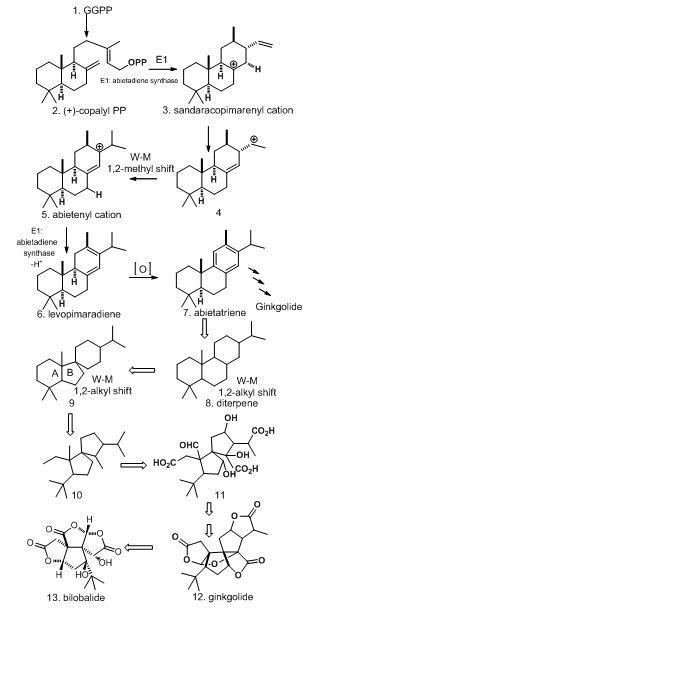
Bilobalide and ginkgolide both have very similar biosynthesis pathway. Bilobalide is formed by partially degraded from ginkgoglide. Bilibalide is derived from geranylgeranyl pyrophosphate (GGPP), which is formed by addition of farnesyl pyrophosphate (FPP) to isopentenyl pyrophosphate (IPP) unit, to form a C15 sesquiterpene. Such formation went through the mevalonate pathway (MVA) and methylerythritol phosphate MEP pathway. In order to generate bilobalide, C20 ginkgolide 13 must form first. To transform from GGPP to abietenyl cation 5, a single bifunctional enzyme abietadiene synthase E1 is required. However, due to the complexity of ginkgolide structures for rearrangement, ring cleavage, and formation of lactone rings, diterpene 8 is used to explain instead. Levopimaradiene 6 and abietatriene 7 are precursors for ginkgolide and bilobalide formation. The unusual tert-butyl substituent is formed from A ring cleavage in 9. Bilobalide 13 then formed in loss of carbons through degradation from ginkgolide 12, and lactones are formed from residual carboxyl and alcohol functions. The end product of bilobalide contains sesquiterpenes and three lactones units.
Pharmacology
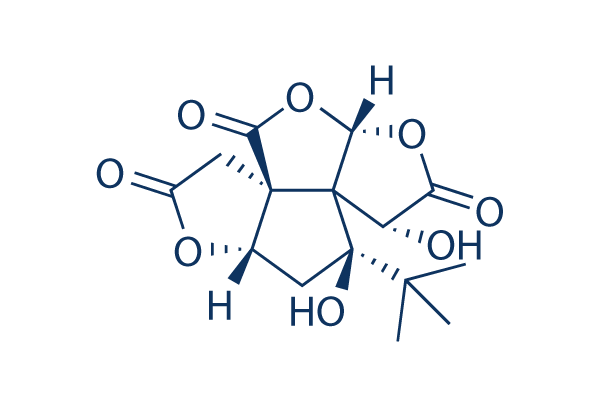
Bilobalide is important for producing several of the effects of Gingko biloba extracts, and it has neuroprotective effects, as well as inducing the liver enzymes CYP3A1 and 1A2, which may be partially responsible for interactions between gingko and other herbal medicines or pharmaceutical drugs. Bilobalide has recently been found to be a negative allosteric modulator at the GABAA and GABAA-rho receptors. Of GABAA, it may possibly be selective for the subunits predominantly implicated in cognitive and memory functioning such as α1.