 | ||
Amyloidosis part 1 definition historical aspects properties of amyloid
Amyloids are aggregates of proteins that become folded into a shape that allows many copies of that protein to stick together forming fibrils. In the human body, amyloids may cause various diseases, as previously healthy proteins lose their normal physiological functions and form fibrous deposits in plaques around cells that disrupt the healthy function of tissues and organs. Such amyloids have been associated with more than 20 human diseases, known as amyloidosis, and may play a role in some neurodegenerative disorders. Some amyloid proteins are infectious; these are called prions in which the infectious form can act as a template to convert other non-infectious proteins into infectious form. Amyloids however may also have normal biological functions; for example, in the formation of fimbriae in some genera of bacteria, transmission of epigenetic traits in fungi, as well as pigment deposition and hormone release in humans.
Contents
- Amyloidosis part 1 definition historical aspects properties of amyloid
- Definition
- Diseases featuring amyloids
- Non disease and functional amyloids
- Structure
- Formation
- Amyloid pathology
- Histological staining
- References
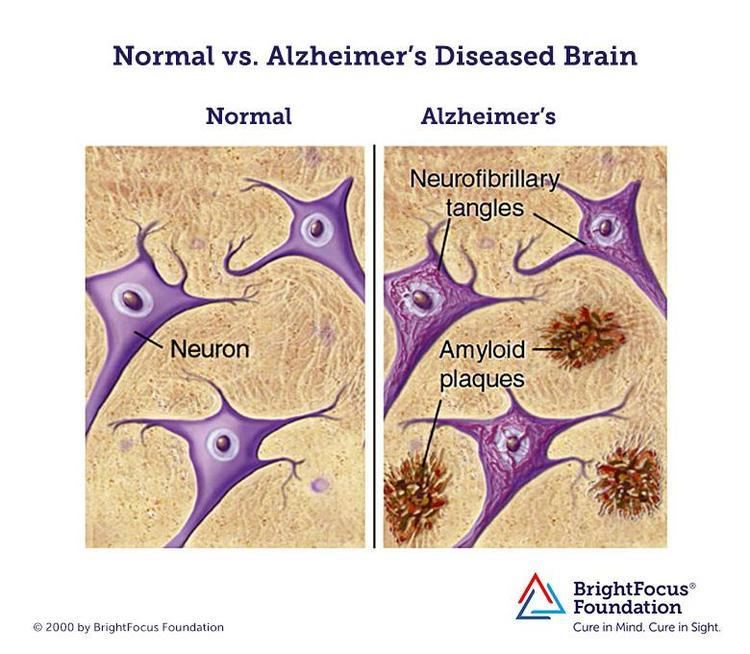
Amyloids have been known to arise from many different proteins and polypeptides. These polypeptide chains generally form β-sheet structures that aggregate into long fibers; however, identical polypeptides can fold into multiple distinct amyloid conformations. The diversity of the conformations may have led to different forms of the prion diseases.
Definition
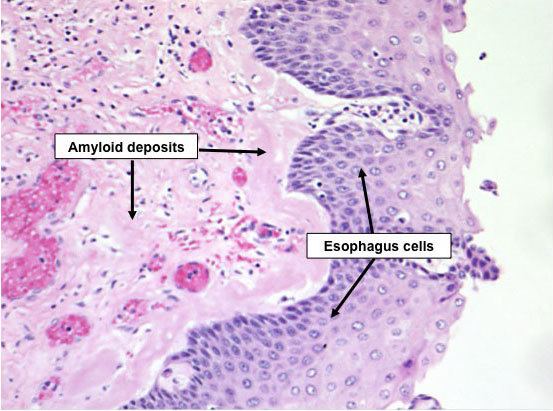
The name amyloid comes from the early mistaken identification by Rudolf Virchow of the substance as starch (amylum in Latin, from Greek ἄμυλον amylon), based on crude iodine-staining techniques. For a period, the scientific community debated whether or not amyloid deposits are fatty deposits or carbohydrate deposits until it was finally found (in 1859) that they are, in fact, deposits of albumoid proteinaceous material.
The remainder of this article will use the biophysical context.
Diseases featuring amyloids
The International Society of Amyloidosis classifies amyloid fibrils based upon associated proteins.
Non-disease and functional amyloids
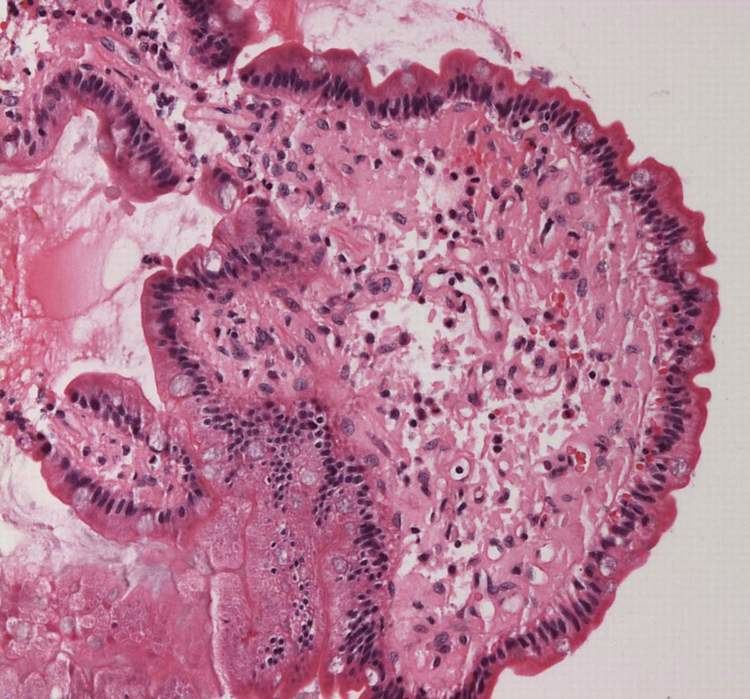
"Amyloid deposits occur in the pancreas of patients with diabetes mellitus, although it is not known if this is functionally important. The major component of pancreatic amyloid is a 37-amino acid residue peptide known as islet amyloid polypeptide or amylin. This is stored with insulin in secretory granules in B cells and is co secreted with insulin" (Rang and Dale's Pharmacology, 2015).
Structure
Amyloid is formed of long unbranched fibers that are characterized by a cross-beta sheet quaternary structure in which parallel chains of β-stranded peptides are arranged in an orientation perpendicular to the axis of the fiber. Each individual fiber may be 5–15 nanometres in width and a few micrometres in length. While amyloid is usually identified using fluorescent dyes, stain polarimetry, circular dichroism, or FTIR (all indirect measurements), the "gold-standard" test to see whether a structure contains cross-beta fibres is by placing a sample in an X-ray diffraction beam. The term "cross-beta" was based on the observation of two sets of diffraction lines, one longitudinal and one transverse, that form a characteristic "cross" pattern. There are two characteristic scattering diffraction signals produced at 4.7 and 10 Ångstroms (0.47 nm and 1.0 nm), corresponding to the interstrand and stacking distances in beta sheets. The "stacks" of beta sheet are short and traverse the breadth of the amyloid fibril; the length of the amyloid fibril is built by aligned strands. The cross-beta pattern is considered a diagnostic hallmark of amyloid structure.
X-ray diffraction studies of microcrystals revealed atomistic details of core region of amyloid. The crystallographic structures show that short stretches from amyloid-prone regions of amyloidogenic proteins run perpendicular to the filament axis, confirming the "cross-beta" model. They also reveal a number of characteristics of amyloid structures – the two layers of beta-sheet are tightly packed together with the interface devoid of water (therefore referred to as dry interface), the opposing β-strands are slightly offset from each other, and their side-chains interdigitate with no hydrogen bond apart from those between stacks of asparagines and glutamines. The compact dehydrated interface created is termed steric-zipper interface. There are eight classes of steric-zipper interfaces, depending on types of beta-sheet (parallel and anti-parallel) and symmetry between two adjacent beta-sheets.
A variety of tertiary structures have been observed in amyloid. The β-sheets may form a β-sandwich, or a β-solenoid which may be either β-helix or β-roll. Identical polypeptides can fold into multiple distinct amyloid conformations.
Formation
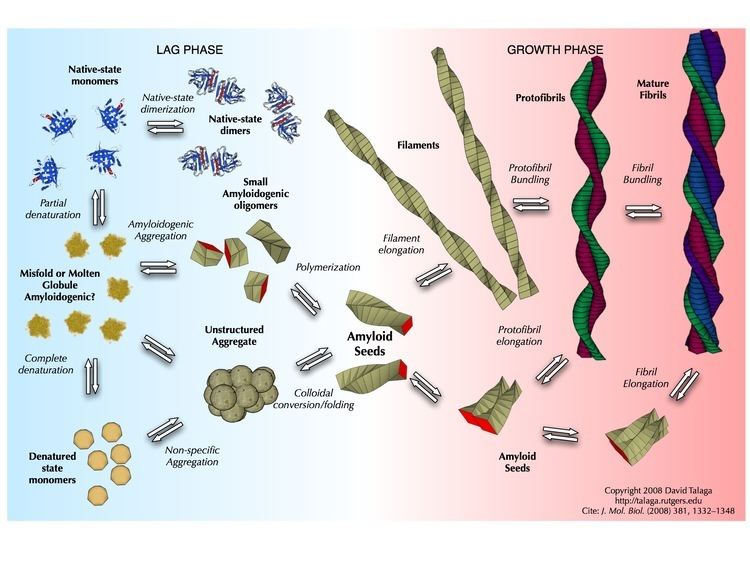
Amyloid is formed through the polymerization of hundreds to thousands of monomeric peptides into long fibers. In general, amyloid polymerization (aggregation or non-covalent polymerization) is sequence-sensitive, that is, causing mutations in the sequence can prevent self-assembly, especially if the mutation is a beta-sheet breaker, such as proline or non-coded alpha-aminoisobutyric acid. For example, humans produce amylin, an amyloidogenic peptide associated with type II diabetes, but in rats and mice prolines are substituted in critical locations and amyloidogenesis does not occur. Studies comparing synthetic to recombinant Amyloid beta 1-42 in assays measuring rate of fibrillation, fibril homogeneity, and cellular toxicity showed that recombinant Amyloid beta 1-42 has a faster fibrillation rate and greater toxicity than synthetic Amyloid beta 1-42 peptide. This observation combined with the irreproducibility of certain Amyloid beta 1-42 experimental studies has been suggested to be responsible for the lack of progress in Alzheimer's research. Consequently, there has been renewed efforts to manufacture Amyloid beta 1-42 and other amyloid peptides at unprecedented (>99%) purity.
There are two broad classes of amyloid-forming polypeptide sequences. Glutamine-rich polypeptides are important in the amyloidogenesis of Yeast and mammalian prions, as well as Trinucleotide repeat disorders including Huntington's disease. When peptides are in a beta-sheet conformation, including arrangements in which the beta-strands are parallel and in-register (causing alignment of residues), glutamines can brace the structure by forming inter-strand hydrogen bonding between its amide carbonyls and nitrogens. In general, for this class of diseases, toxicity correlates with glutamine content. This has been observed in studies of onset age for Huntington's disease (the longer the polyglutamine sequence the sooner the symptoms appear), and has been confirmed in a C. elegans model system with engineered polyglutamine peptides.
Other polypeptides and proteins such as amylin and the Alzheimer's beta protein do not have a simple consensus sequence and are thought to operate by hydrophobic association. Among the hydrophobic residues, aromatic amino-acids are found to have the highest amyloidogenic propensity.
For these peptides, cross-polymerization (fibrils of one polypeptide sequence causing other fibrils of another sequence to form) is observed in vitro and possibly in vivo. This phenomenon is important, since it would explain interspecies prion propagation and differential rates of prion propagation, as well as a statistical link between Alzheimer's and type 2 diabetes. In general, the more similar the peptide sequence the more efficient cross-polymerization is, though entirely dissimilar sequences can cross-polymerize and highly similar sequences can even be "blockers" that prevent polymerization. Polypeptides will not cross-polymerize their mirror-image counterparts, indicating that the phenomenon involves specific binding and recognition events.
The fast aggregation process, rapid conformational changes as well as solvent effects provide challenges in measuring monomeric and oligomeric amyloid peptide structures in solution. Theoretical and computational studies complement experiments and provide insights that are otherwise difficult to obtain using conventional experimental tools. Several groups have successfully studied the disordered structures of amyloid and reported random coil structures with specific structuring of monomeric and oligomeric amyloid as well as how genetics and oxidative stress impact the flexible structures of amyloid in solution.
Oligomeric intermediates of insulin during fibrillation (more toxic than other intermediates: native, protofibril, and fibril) decreased the surface tension of solution which indicated to detergent-like properties of oligomers and significant role of hydrophobic forces in cytotoxicity of oligomers.
Amyloid pathology
The reasons for amyloid association disease are unclear. In some cases, the deposits physically disrupt tissue architecture, suggesting disruption of function by some bulk process. An emerging consensus implicates prefibrillar intermediates rather than mature amyloid fibers in causing cell death.
Calcium dysregulation has been observed in cells exposed to amyloid oligomers. These small aggregates can form ion channels planar lipid bilayer membranes. Channel formation has been hypothesized to account for calcium dysregulation and mitochondrial dysfunction by allowing indiscriminate leakage of ions across cell membranes.
Studies have shown that amyloid deposition is associated with mitochondrial dysfunction and a resulting generation of reactive oxygen species (ROS), which can initiate a signalling pathway leading to apoptosis.
There are reports that indicate amyloid polymers (such as those of huntingtin, associated with Huntington's disease) can induce the polymerization of essential amyloidogenic proteins, which should be deleterious to cells. Also, interaction partners of these essential proteins can also be sequestered.
Histological staining
In the clinical setting, amyloid diseases are typically identified by a change in the fluorescence intensity of planar aromatic dyes such as thioflavin T, congo red or NIAD-4. In general, this is attributed to the environmental change, as these dyes intercalate between beta-strands to confine their structure. Congo Red positivity remains the gold standard for diagnosis of amyloidosis. In general, binding of Congo Red to amyloid plaques produces a typical apple-green birefringence when viewed under cross-polarized light. To avoid nonspecific staining, other histology stains, such as the hematoxylin and eosin stain, are used to quench the dyes' activity in other places such as the nucleus, where the dye might bind. Modern antibody technology and immunohistochemistry has made specific staining easier, but often this can cause trouble because epitopes can be concealed in the amyloid fold; in general, an amyloid protein structure is a different conformation from the one that the antibody recognizes.
