Formula C6H7N Density 943 kg/m³ | Molar mass 93.13 g/mol | |
 | ||
Appearance Faintly yellow-green clear liquid | ||
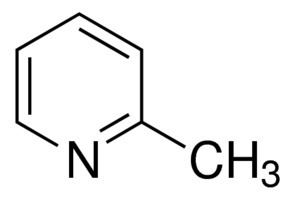
2-Methylpyridine, or 2-picoline, is the compound described with formula C6H7N. 2-Picoline is a colorless liquid that has an unpleasant odor similar to pyridine. It is mainly used to make vinylpyridine and the agrichemical nitrapyrin.
Contents
Synthesis
2-Picoline was the first pyridine compound reported to be isolated in pure form. It was isolated from coal tar in 1846 by T. Anderson. It is now mainly produced by two principal routes, the condensation of acetaldehyde, formaldehyde, and ammonia and the cyclization of nitriles and acetylene. One example of such reaction is the combination of acetaldehyde and ammonia:
Approximately 8000 t/a was produced worldwide in 1989.
Reactions
Most of the reactions of picoline are centered on the methyl group. For example, the principal use of 2-picoline is as a precursor of 2-vinylpyridine. The conversion is achieved by condensation with formaldehyde:
The copolymer of 2-vinylpyridine, butadiene and styrene is used as an adhesive for textile tire cord. 2-Picoline is also a precursor to the agrichemical, nitrapyrin, which prevents loss of ammonia from fertilizers. Oxidation by potassium permanganate affords picolinic acid:
Deprotonation with butyllithium affords C5H4NCH2Li, a versatile nucleophile.
N-alkylation of picoline is used in production of amprolium, an antiprotozoan.
Biodegradation
Like other pyridine derivatives, 2-methylpyridine is often reported as an environmental contaminant associated with facilities processing oil shale or coal, and has also been found at legacy wood treatment sites. The compound is readily degradable by certain microorganisms, such as Arthrobacter sp. strain R1 (ATTC strain number 49987), which was isolated from an aquifer contaminated with a complex mixture of pyridine derivatives. Arthrobacter and closely related Actinobacteria are often found associated with degradation of pyridine derivatives and other nitrogen heterocyclic compounds. 2-methypyridine and 4-methypyridine are more readily degraded and exhibit less volatilization loss from environmental samples than does 3-methypyridine.
