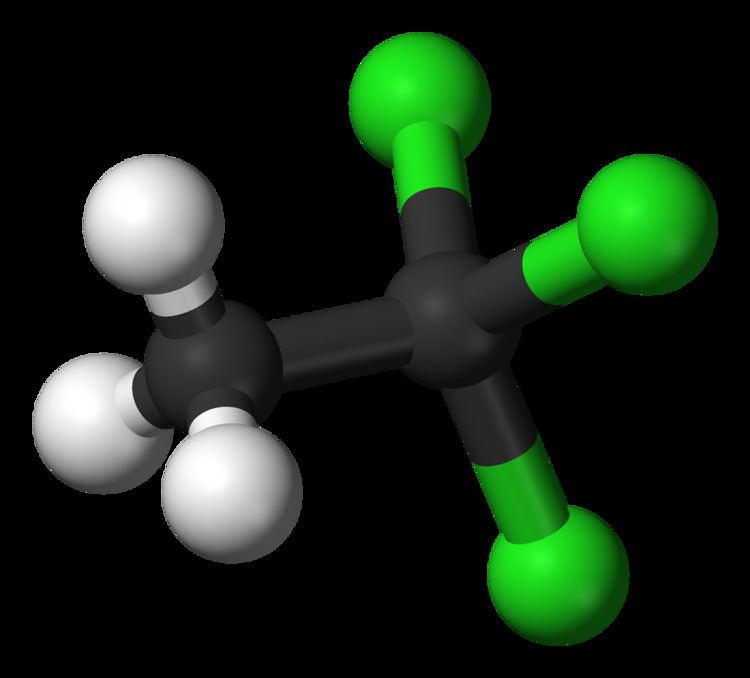
Appearance Colorless liquid Molar mass 133.4 g/mol | Formula C2H3Cl3 Density 1.32 g/cm³ | |
 | ||
The organic compound 1,1,1-trichloroethane, also known as methyl chloroform, is a chloroalkane. This colourless, sweet-smelling liquid was once produced industrially in large quantities for use as a solvent. It is regulated by the Montreal Protocol as an ozone-depleting substance and its use is being rapidly phased out.
Contents
Production

1,1,1-Trichloroethane was first reported by Henri Victor Regnault in 1840. Industrially, it is usually produced in a two-step process from vinyl chloride. In the first step, vinyl chloride reacts with hydrogen chloride at 20-50 °C to produce 1,1-dichloroethane:
CH2=CHCl + HCl → CH3CHCl2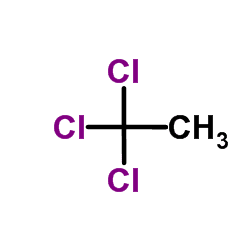
This reaction is catalyzed by a variety of Lewis acids, mainly aluminium chloride, iron(III) chloride, or zinc chloride. The 1,1-dichloroethane is then converted to 1,1,1-trichloroethane by reaction with chlorine under ultraviolet irradiation:
CH3CHCl2 + Cl2 → CH3CCl3 + HCl
This reaction proceeds at 80-90% yield, and the hydrogen chloride byproduct can be recycled to the first step in the process. The major side-product is the related compound 1,1,2-trichloroethane, from which the 1,1,1-trichloroethane can be separated by distillation.

A somewhat smaller amount of 1,1,1-trichloroethane is produced from the reaction of vinylidene chloride and hydrogen chloride in the presence of an iron(III) chloride catalyst:
CH2=CCl2 + HCl → CH3CCl31,1,1-Trichloroethane is marketed with stabilizers since it is unstable with respect to dehydrochlorination and attacks some metals. Stabilizers comprise up to 8% of the formulation, including acid scavengers (epoxides, amines) and complexants. The Montreal Protocol targeted 1,1,1-trichloroethane as one of those compounds responsible for ozone depletion and banned its use beginning in 1996. Since then, its manufacture and use has been phased out throughout most of the world.
Uses
1,1,1-trichloroethane is generally considered a non-polar solvent. Owing to the good polarizability of the chlorine atoms, it is a superior solvent for organic compounds that do not dissolve well in hydrocarbons such as hexane. It is an excellent solvent for many organic materials and also one of the least toxic of the chlorinated hydrocarbons. Prior to the Montreal Protocol, it was widely used for cleaning metal parts and circuit boards, as a photoresist solvent in the electronics industry, as an aerosol propellant, as a cutting fluid additive, and as a solvent for inks, paints, adhesives and other coatings. 1,1,1-trichloroethane is also used as an insecticidal fumigant.
It was also the standard cleaner for photographic film (movie/slide/negatives, etc.). Other commonly available solvents damage emulsion, and thus are not suitable for this application. The standard replacement, Forane 141 is much less effective, and tends to leave a residue. 1,1,1-trichloroethane was used as a thinner in correction fluid products such as liquid paper. Many of its applications previously used carbon tetrachloride (which was banned in US consumer products in 1970). In turn, 1,1,1-trichloroethane itself is now being replaced by other solvents in the laboratory.
Safety
Although not as toxic as many similar compounds, inhaled or ingested 1,1,1-trichloroethane does act as a central nervous system depressant and can cause effects similar to those of ethanol intoxication, including dizziness, confusion, and in sufficiently high concentrations, unconsciousness and death. Fatal poisonings and illnesses linked to intentional inhalation of trichloroethane have been reported. The removal of the chemical from correction fluid commenced due to Proposition 65 declaring it hazardous and toxic
Prolonged skin contact with the liquid can result in the removal of fats from the skin, resulting in chronic skin irritation. Studies on laboratory animals have shown that 1,1,1-trichloroethane is not retained in the body for long periods of time. However, chronic exposure has been linked to abnormalities in the liver, kidneys, and heart. Pregnant women should avoid exposure, as the compound has been linked to birth defects in laboratory animals (see teratogenesis).
