Formula C4H7N | ||
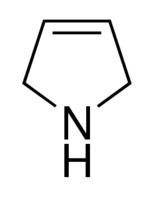 | ||
Pyrrolines, also known under the name dihydropyrroles, are three different heterocyclic organic chemical compounds that differ in the position of the double bond. Pyrrolines are formally derived from the aromate pyrrole by hydrogenation. 1-Pyrroline is a cyclic imine, whereas 2-pyrroline and 3-pyrroline are cyclic amines.
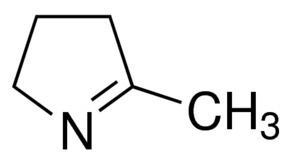
Substituted pyrrolines
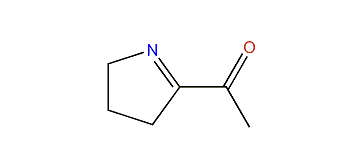
Notable examples of pyrrolines containing various substituents include:
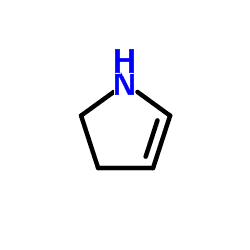
N-substituted pyrrolines can be generated by ring-closing metathesis.
References
Pyrroline Wikipedia(Text) CC BY-SA
