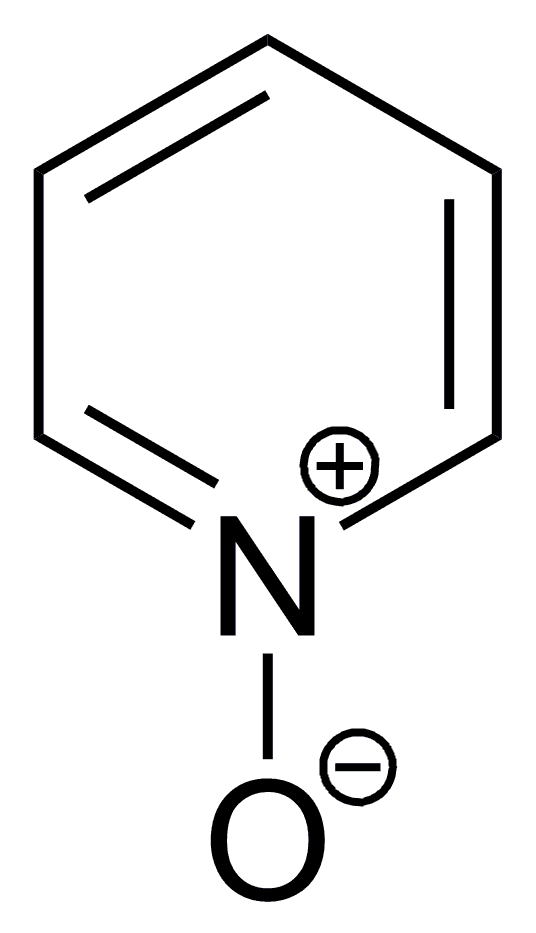
Formula C5H5NO | Appearance colourless solid | |
 | ||
Pyridine N-oxide: Basic concept and preparation with complete mechanistic description.
Pyridine-N-oxide is the heterocyclic compound with the formula C5H5NO. This colourless, hygroscopic solid is the product of the oxidation of pyridine. It was originally prepared using peracids as the oxidising agent. The molecule is planar. The compound is used infrequently as an oxidizing reagent in organic synthesis. It also serves as a ligand in coordination chemistry, and to activate the pyridine ring towards aromatic electrophilic substitution.
Contents
- Pyridine N oxide Basic concept and preparation with complete mechanistic description
- Synthesis and reactions
- Safety
- References
Synthesis and reactions
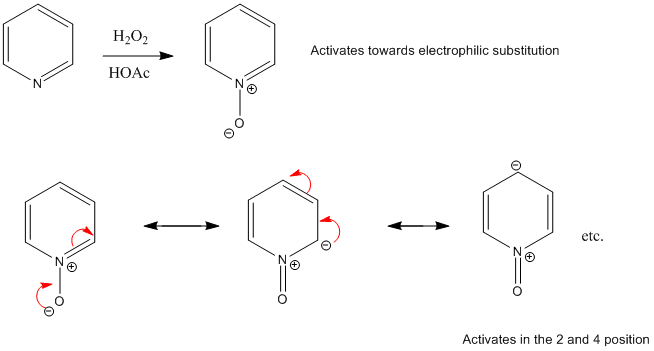
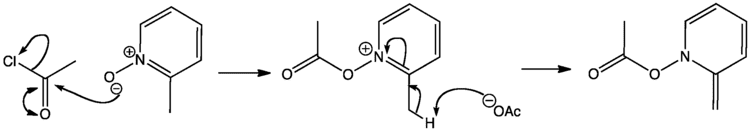
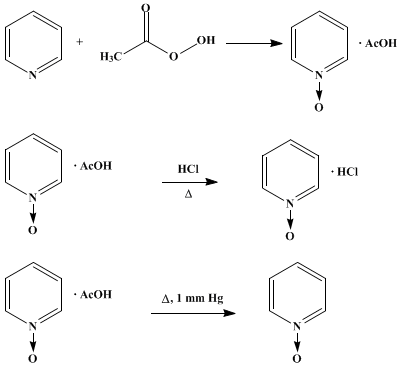
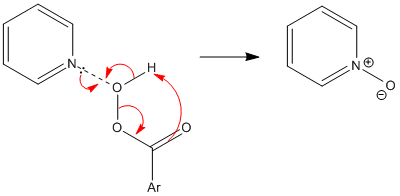
The oxidation of pyridine can be achieved with a number of peracids including peracetic acid and perbenzoic acid in a reaction that affords the protonated derivative. Subsequent treatment with heating under low pressure removes acids as gases to liberates the neutral oxide. More recent reported approaches include a modified Dakin reaction using a urea-hydrogen peroxide complex, and oxidation by sodium perborate or, using methylrhenium trioxide (CH
3ReO
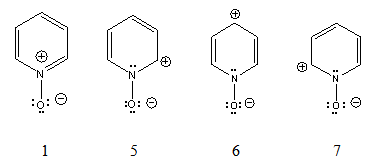
3) as catalyst, with sodium percarbonate. Pyridine N-oxide is five orders of magnitude less basic than pyridine, but it is isolable as a hydrochloride salt, [C5H5NOH]Cl, via a synthesis reported in Organic Syntheses:
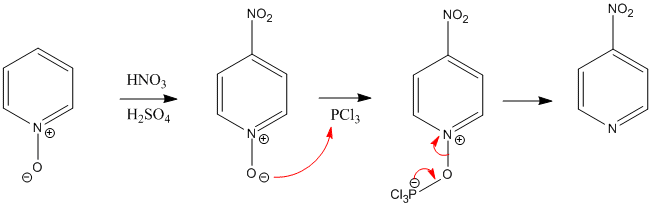
Considerable work has been done on synthesis of N-oxides from substituted pyridine starting materials and on chemical modifications of amine oxide systems, including the chlorination of the pyridine-N-oxide with phosphorus oxychloride gives 4- and 2-chloropyridines.
Safety
The compound is a skin irritant.