Formula KO2 Appearance yellow solid | Density 2.14 g/cm³ | |
 | ||
Alkali metals 18 reaction of potassium superoxide with lithium
Potassium superoxide is the inorganic compound with the formula KO
2. It is a yellow paramagnetic solid that decomposes in moist air. It is a rare example of a stable salt of the superoxide ion. Potassium superoxide is used as a CO
2 scrubber, H
2O dehumidifier and O
2 generator in rebreathers, spacecraft, submarines and spacesuit life support systems.
Contents
- Alkali metals 18 reaction of potassium superoxide with lithium
- Production and reactions
- Reactivity
- Applications
- Hazards
- References
Production and reactions
Potassium superoxide is produced by burning molten potassium in an atmosphere of oxygen.
K + O2 → KO
2
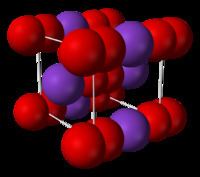
The salt consists of K+
and O−
2 ions, linked by ionic bonds. The O-O distance is 1.28 Å.
Reactivity
Hydrolysis gives oxygen gas, hydrogen peroxide and potassium hydroxide:
2 KO2 + 2H
2O → 2 KOH + H
2O
2 + O
2
Its degradation by carbon dioxide affords carbonates:
4 KOH + 2 CO2 → 2 K2CO3 + 2 H2OCombinations of these two reactions occur as well:

2 + 2 CO2 → 2 K2CO3 + 3 O
24 KO
2 + 4 CO2 + 2 H2O → 4 KHCO3 + 3 O
2
Potassium superoxide finds only niche uses as a laboratory reagent. Because it reacts with water, KO
2 is often studied in organic solvents. Since the salt is poorly soluble in nonpolar solvents, crown ethers are typically used. The tetraethylammonium salt is also known. Representative reactions of these salts involve the use of superoxide as a nucleophile, e.g., in the conversion of alkyl bromides to alcohols and acyl chlorides into diacyl peroxides.
Applications
The Russian Space Agency has had success using potassium superoxide in chemical oxygen generators for its spacesuits and Soyuz spacecraft. KO
2 has also been utilized in canisters for rebreathers for fire fighting and mine rescue work, but had limited use in scuba rebreathers because of its dangerously explosive reaction with water. The theoretical capacity of KO
2 is the absorption of 0.618 kg CO2 per kg of absorbent while 0.450 kg O
2 are generated per kg of absorbent. For one KO
2 molecule, it's one CO2 molecule but only 0.75 oxygen molecules. The human body though will produce less CO2 molecules than oxygen molecules needed because oxidation of food also needs oxygen to produce water and urea.
Hazards
Potassium superoxide is a potent oxidizer, and can produce explosive reactions when combined with a variety of substances, including water, acids, organics, or powdered graphite. Even dry superoxide can produce an impact-sensitive explosive compound when combined with organic oils such as kerosene. In 1999 at Oak Ridge National Laboratory, cleanup of potassium oxides from a NaK metal leak produced an impact-sensitive explosion while saturated with mineral oil.

