Related compounds Molar mass 136.169 g/mol Melting point 197 °C | Formula KHSO4 Density 2.24 g/cm³ Appearance colorless solid | |
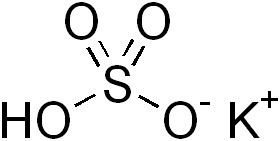 | ||
Potassium bisulfate is an inorganic compound with the chemical formula KHSO4 and is the potassium acid salt of sulfuric acid.
Contents
Natural Occurrence
Mercallite, the mineralogical form of potassium bisulfate, occurs very rarely. Misenite is another, more complex, form of potassium bisulfate.
Preparation
Potassium bisulfate is prepared by neutralizing sulfuric acid with an equal molar amount of a potassium containing base, for example potassium hydroxide:
H2SO4 + KOH → KHSO4 + H2OPotassium bisulfate is also formed by the union of sulfuric acid with potassium sulfate:
H2SO4 + K2SO4 → 2 KHSO4Potassium bisulfate is also the main by-product in the production of nitric acid from potassium nitrate and sulfuric acid:
KNO3 + H2SO4 → KHSO4 + HNO3Chemical Properties
Thermal decomposition of potassium bisulfate forms potassium pyrosulfate and water:
2 KHSO4 → K2S2O7 + H2OTemperatures above 600 °C further decompose potassium bisulfate to potassium sulfate and sulfur trioxide:
2 KHSO4 → K2SO4 + SO3 + H2OAqueous solutions of potassium bisulfate behave as two separate, uncombined compounds, K2SO4 and H2SO4. Adding ethanol to the solution precipitates out potassium sulfate.
Uses
Potassium bisulfate is commonly used to prepare potassium bitartrate for winemaking. Potassium bisulfate is also used as a disintegrating agent in analytical chemistry or as a precursor to prepare potassium persulfate, a powerful oxidizing agent.
