Formula C13H10O3 Density 1.25 g/cm³ Molar mass 214.22 g/mol | Melting point 41.5 °C Boiling point 173 °C Appearance White solid | |
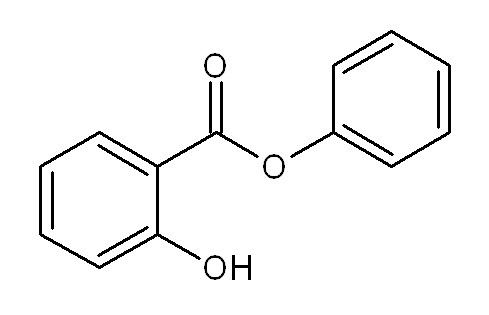 | ||
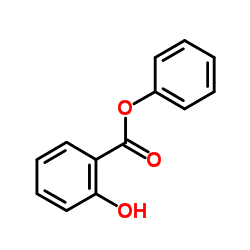
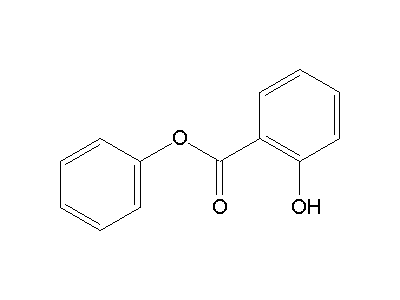
Phenyl salicylate
Phenyl salicylate, or salol, is a chemical substance, introduced in 1886 by Marceli Nencki of Basel. It can be created by heating salicylic acid with phenol. Once used in sunscreens, phenyl salicylate is now used in the manufacture of some polymers, lacquers, adhesives, waxes and polishes. It is also used frequently in school laboratory demonstrations on how cooling rates affect crystal size in igneous rocks.
Contents
Phenyl salicylate crystal growth salol
Salol reaction
In the salol reaction, phenyl salicylate reacts with o-toluidine in 1,2,4-trichlorobenzene at elevated temperatures to the corresponding amide o-Salicylotoluide. Salicylamides are one type of drug.
Medical
It has been used as an antiseptic based on the antibacterial activity upon hydrolysis in the small intestine.
It acts as a mild analgesic.

References
Phenyl salicylate Wikipedia(Text) CC BY-SA
