 | ||
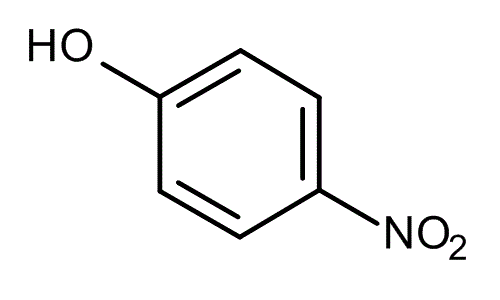
Nitrophenols consist of a phenol molecule with one or more nitro-groups attached to the aromatic ring. The term is most often used to describe singly nitrated phenols.
Contents
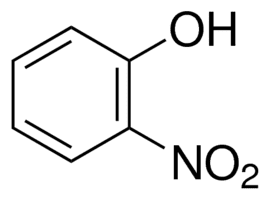
Lower pka p nitrophenol or m nitrophenol
Mono-nitrophenols
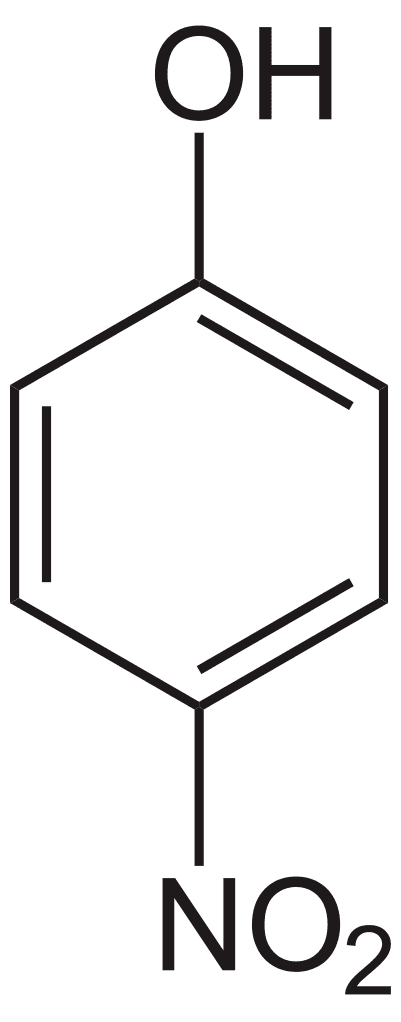
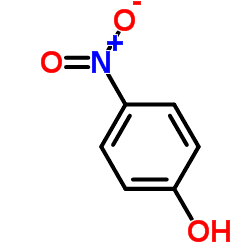
with the formula HOC6H4NO2. Three isomeric nitrophenols exist:
The mononitrated phenols are often hydrogenated to the corresponding aminophenols that are also useful industrially.
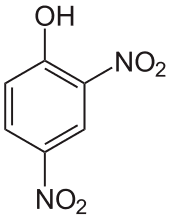
Di- and trinitrophenols
2,4-Dinitrophenol (m.p. 83 °C) is a moderately strong acid (pKa = 4.89). 2,4,6-trinitrophenol is better known as picric acid, which has a well-developed chemistry.
Safety
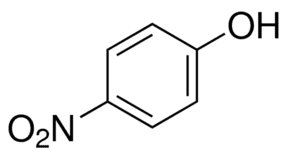
Nitrophenols are poisonous. Occasionally, nitrophenols contaminate the soil near former explosives or fabric factories and military plants, and current research is aimed at remediation.
References
Nitrophenol Wikipedia(Text) CC BY-SA
