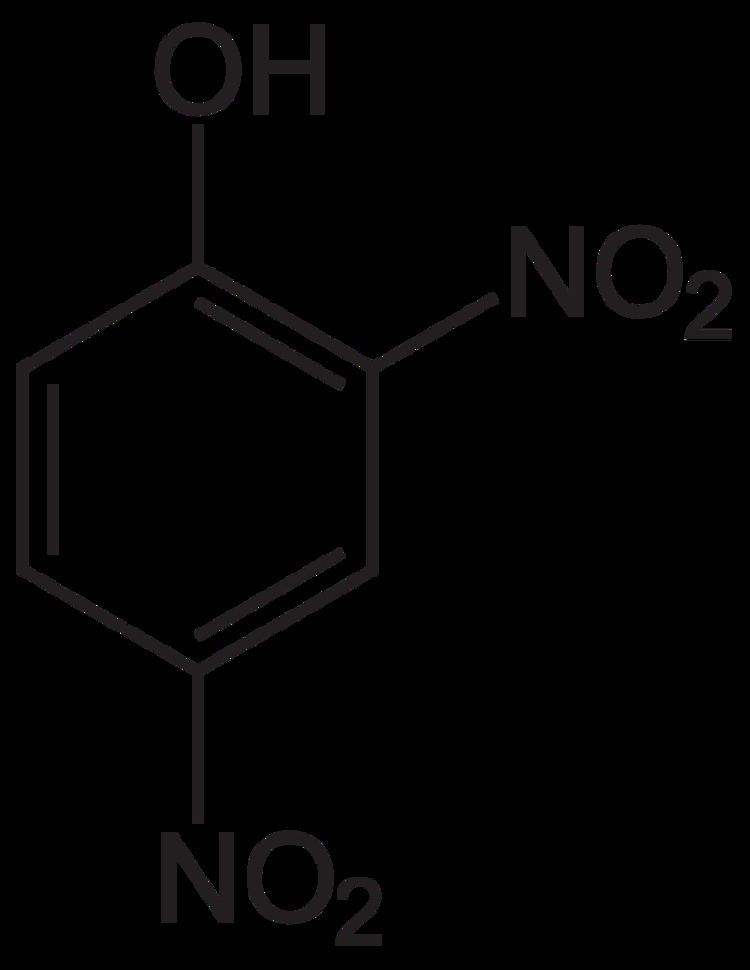
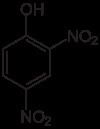
Formula C6H4N2O5 Molar mass 184.106 g/mol Boiling point 113 °C | IUPAC ID 2,4-dinitrophenol Melting point 108 °C Density 1.68 g/cm³ | |
 | ||
Soluble in Benzene, Ethyl acetate, Toluene, Pyridine, Alcohol, Carbon tetrachloride, Acetone, Chloroform | ||
Chemcast ep2 2 4 dinitrophenol dnp
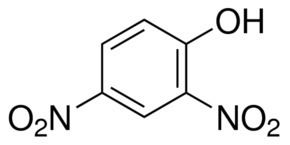
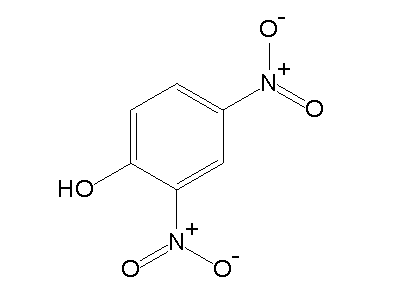
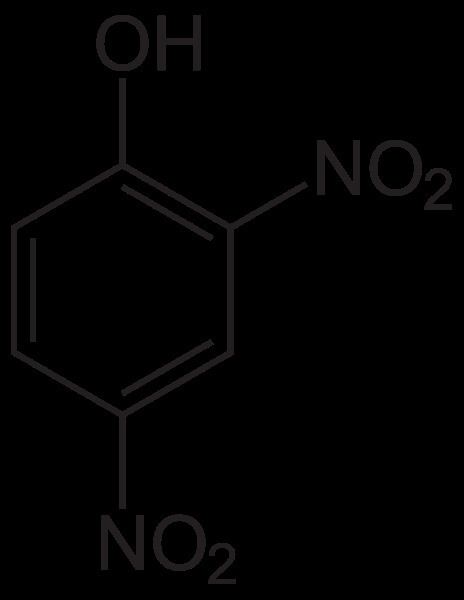
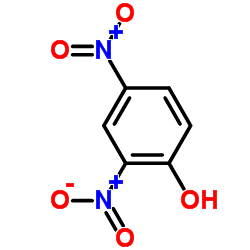
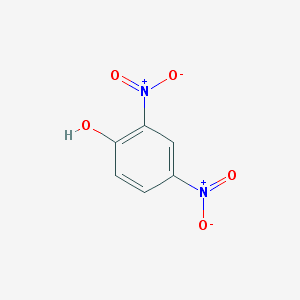
2,4-Dinitrophenol (2,4-DNP or simply DNP) is an organic compound with the formula HOC6H3(NO2)2. It is a yellow, crystalline solid that has a sweet, musty odor. It sublimes, is volatile with steam, and is soluble in most organic solvents as well as aqueous alkaline solutions. It is a precursor to other chemicals and is biochemically active, inhibiting energy (adenosine triphosphate, ATP) production in cells with mitochondria. Its use in high doses as a dieting aid has been identified with severe side-effects, including a number of deaths.
Contents
- Chemcast ep2 2 4 dinitrophenol dnp
- Synthesis
- Uses
- Biochemical aspects
- Health effects
- Dieting aid
- Pharmacokinetics
- Commerce
- Environmental effects
- References

Synthesis
DNP is produced by hydrolysis of 1-chloro-2,4-dinitrobenzene.
Uses
Commercial DNP is used as an antiseptic and as a pesticide. It is a chemical intermediate in the production of sulfur dyes, wood preservatives, and some herbicides including dinoseb and dinoterb. DNP has also been used to make photographic developers and explosives.
Biochemical aspects
In living cells, DNP acts as a proton ionophore, an agent that can shuttle protons (hydrogen cations) across biological membranes. It dissipates the proton gradient across mitochondria and chloroplast membranes, collapsing the proton motive force that the cell uses to produce most of its ATP chemical energy. Instead of producing ATP, the energy of the proton gradient is lost as heat.

DNP is often used in biochemistry research to help explore the bioenergetics of chemiosmotic and other membrane transport processes.
Health effects
DNP is considered to have high acute toxicity. Acute oral exposure to DNP has resulted in increased basal metabolic rate, nausea, vomiting, sweating, dizziness, headache, and loss of weight. Chronic oral exposure to DNP can lead to the formation of cataracts and skin lesions and has caused effects on the bone marrow, central nervous system, and cardiovascular system.
Dieting aid
DNP was used extensively in diet pills from 1933 to 1938 after Cutting and Tainter at Stanford University made their first report on the drug's ability to greatly increase metabolic rate. After only its first year on the market Tainter estimated that at least 100,000 persons had been treated with DNP in the United States, in addition to many others abroad. DNP acts as a protonophore, allowing protons to leak across the inner mitochondrial membrane and thus bypass ATP synthase. This makes ATP energy production less efficient. In effect, part of the energy that is normally produced from cellular respiration is wasted as heat. The inefficiency is proportional to the dose of DNP that is taken. As the dose increases and energy production is made more inefficient, metabolic rate increases (and more fat is burned) in order to compensate for the inefficiency and to meet energy demands. DNP is probably the best known agent for uncoupling oxidative phosphorylation. The "phosphorylation" of adenosine diphosphate (ADP) by ATP synthase gets disconnected or "uncoupled" from oxidation.
The factor that limits ever-increasing doses of DNP is not a lack of ATP energy production, but rather an excessive rise in body temperature due to the heat produced during uncoupling. Accordingly, DNP overdose will cause fatal hyperthermia, with body temperature rising to as high as 44 °C (111 °F) shortly before death. In light of this, when it was used clinically, the dose was slowly titrated according to personal tolerance, which varies greatly.
Case reports have shown that an acute administration of 20–50 mg per kilogram of body weight in humans can be lethal. Concerns about dangerous side-effects and rapidly developing cataracts resulted in DNP being discontinued in the United States by the end of 1938. DNP, however, continues to be used by some bodybuilders and athletes to rapidly lose body fat. Fatal overdoses include cases of accidental exposure, suicide, and excessive intentional exposure (overdose). The substance's use as a dieting aid has also led to a number of accidental fatalities.
Although DNP is widely considered too dangerous for clinical use due to its numerous potential side effects, such as cataracts, its mechanism of action remains under investigation as a potential approach for treating obesity. Currently, research is being conducted on uncoupling proteins naturally found in humans.
The United Kingdom's Food Standards Agency identifies DNP as "an industrial chemical known to have serious short-term and long-term effects, which can be extremely dangerous to human health." and advises "consumers not to take any product containing DNP at any level. This chemical is not suitable for human consumption."
Pharmacokinetics
Information about pharmacokinetics of DNP in humans is limited and conflicting. The EPA states that "Data on the elimination kinetics of the dinitrophenols or their metabolic products in humans were not found." The ATSDR's Toxicological Profile for Dinitrophenols also states that "No studies were located regarding distribution in humans after oral exposure to 2,4-DNP. Limited information is available regarding distribution in animals after oral exposure to 2,4-DNP." However, they do state that "Elimination from the body appears to be rapid, except possibly in cases of compromised liver function." This coincides with a review in the NEJM on the biological actions of dinitrophenol, which stated that "Judging from the metabolic response, DNP appears to be eliminated entirely in three or four days; in the presence of liver or kidney damage it is possible that the drug will be retained over a longer period." Oddly, more recent papers give an array of possible half-lives, ranging from 3 hours, to 5–14 days. Other recent papers maintain that the half-life in humans is unknown.
Although further investigation is needed, one case report notes that dinitrophenol-induced hyperthermia has been successfully resolved with dantrolene administration. "Dinitrophenol uncouples oxidative phosphorylation, causes release of calcium from mitochondrial stores and prevents calcium re-uptake. This leads to free intracellular calcium and causes muscle contraction and hyperthermia. Dantrolene inhibits calcium release from the sarcoplasmic reticulum which reduces intracellular calcium. The resulting muscle relaxation allows heat dissipation. There is little risk to dantrolene administration. Since dantrolene may be effective in reducing hyperthermia caused by agents that inhibit oxidative phosphorylation, early administration may improve outcome."
Commerce
In 2003, a vendor of DNP was sentenced to prison for mail fraud, with the FDA's OCI investigators having gathered evidence that the vendor's encapsulation of DNP was neither accurate nor sanitary.
Environmental effects
DNP is considered an important environmental contaminant by the United States Environmental Protection Agency. It has been found in 61 of 1400 priority sites that need clean-up of industrial waste. It can enter the air from automobile exhaust, burning of certain industrial substances, and from reaction of nitrogen in air with other atmospheric chemicals. The major site of degradation is the soil, where microorganisms metabolize it.
However, the effects of DNP on anaerobic micro-organisms are still largely undetermined. Some studies suggest that there is anaerobic toxicity due to a reduced methane production.
DNP forms explosive salts with strong bases and ammonia, and emits toxic fumes of nitrogen dioxide when heated to decomposition.
